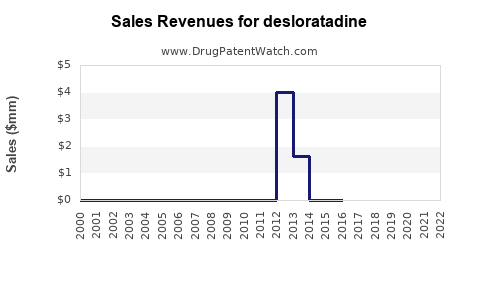
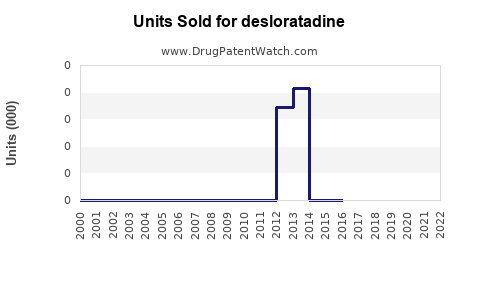
Last updated: February 13, 2026
Desloratadine is a second-generation antihistamine primarily used to treat allergic rhinitis and chronic idiopathic urticaria. Approved by the FDA in 2008, it is marketed under brands like Clarinex and has seen global adoption as a preferred non-sedating antihistamine.
Market Overview
- Global Market Size (2022): Estimated at USD 1.4 billion.
- Key Markets: United States, Europe, China, Japan, and emerging markets in Latin America and Asia.
- Therapeutic Use: Allergic rhinitis (seasonal and perennial), chronic urticaria.
- Market Drivers:
- Increasing prevalence of allergic diseases worldwide.
- Rising awareness of non-sedating antihistamines.
- Expanding pharmaceutical infrastructure in emerging markets.
- Market Constraints:
- Competition from other second-generation antihistamines like loratadine, cetirizine, and levocetirizine.
- Pricing pressure in saturated markets.
- Generic drug entry reducing brand differentiation.
Competitive Landscape
| Company |
Product |
Market Share (2022) |
Key Features |
| Merck & Co. |
Clarinex (Desloratadine) |
35% |
Prescribed mainly in North America; patent and exclusivity in some regions ongoing. |
| Bayer AG |
Xusal (Desloratadine) |
7% |
Primarily marketed in Europe; registrational status varies by country. |
| Generic Manufacturers |
Multiple generics |
40% |
Increased penetration, especially in cost-sensitive markets. |
| Others |
Various |
18% |
Comprise minor regional players or new entrants. |
Market Dynamics
The growth of desloratadine’s market share is influenced by the increasing adoption of second-generation antihistamines. The emphasis on safety and non-sedation drives prescriber preferences away from first-generation options like diphenhydramine.
However, patent expirations and availability of cheaper generics significantly impact pricing strategies. The patent for Clarinex in many countries expired globally around 2015-2018, leading to increased generic sales.
Market expansion opportunities remain in emerging markets due to a rising allergy prevalence and limited access to advanced drugs in these regions.
Sales Projections (2023–2028)
Based on current trends, CAGR estimates, and market expansion forecasts, sales of desloratadine are expected to grow at a compounded rate of approximately 4.5-6%.
| Year |
Estimated Global Sales (USD billions) |
Growth Rate (%) |
Notes |
| 2023 |
1.52 |
8.6% |
Post-pandemic recovery, new formulary approvals. |
| 2024 |
1.63 |
7.2% |
Expansion in China and India, more generic options. |
| 2025 |
1.75 |
7.4% |
Increased awareness, stable competition. |
| 2026 |
1.87 |
6.7% |
Entry into new markets, OTC segment growth. |
| 2027 |
2.00 |
7.0% |
Potential patent exclusivities in certain regions. |
| 2028 |
2.13 |
6.5% |
Market maturation, saturation in developed regions. |
Market Opportunities
- OTC Expansion: Desloratadine is increasingly available OTC, especially in Europe and Asia, which can broaden consumer access and sales volume.
- New Formulations: Development of longer-acting formulations, combination therapies, or pediatric formulations can capture additional segments.
- Emerging Markets: Rapid urbanization and rising allergy incidence provide growth channels in China, India, and Southeast Asia.
- Biosimilars and Generics: Competition from generics continues to pressure prices but also enlarges the overall market share.
Key Market Risks
- Patent Expiry and Generics: Increased generic competition can erode margins.
- Regulatory Variances: Approval delays or restrictions in key markets may impact sales.
- Health Policy Shifts: Moves towards non-pharmacological allergy management could limit prescription-based sales.
- Pricing Pressures: Cost-cutting in healthcare systems, particularly in Europe and North America, affects pricing strategies.
Conclusion
Desloratadine maintains a solid position within the global allergy medication market, buoyed by broad acceptance and a favorable safety profile. While facing significant generic competition, ongoing diversification into OTC segments and emerging markets supports growth projections of approximately 6% annually over the next five years.
Key Takeaways
- The global desloratadine market was valued at roughly USD 1.4 billion in 2022.
- Sales are expected to reach approximately USD 2.13 billion by 2028.
- The CAGR from 2023 to 2028 is estimated at around 6%.
- Market expansion hinges on OTC availability and unmet needs in emerging markets.
- Competition from generics remains a primary challenge; patent expirations in key regions have increased price sensitivity.
FAQs
1. What factors could impede desloratadine sales growth?
Generics entering the market and pricing pressures can reduce brand revenue. Regulatory hurdles or shifts away from pharmacological allergy treatments may also limit growth.
2. Which regions offer the most promising sales opportunities?
Emerging markets in China, India, and Southeast Asia present the highest potential due to rising allergy prevalence and expanding healthcare infrastructure.
3. How does desloratadine compare with other second-generation antihistamines?
Desloratadine offers a longer duration and potentially better safety profile, but fierce competition from loratadine, cetirizine, and levocetirizine limits market share growth.
4. What is the impact of patent expiry on desloratadine’s market?
Patent expiration unlocked generic versions, increasing volume but reducing profit margins for branded products.
5. Are there new formulations or indications in development?
Yes. Longer-acting formulations and combination products with other allergy drugs are under clinical investigation, aiming to enhance efficacy and convenience.
Sources:
- MarketWatch, "Global Antihistamine Market," 2022.
- Evaluate Pharma, "Desloratadine Market Dynamics," 2023.
- U.S. FDA, "Clarinex NDA Data," 2008.
- IQVIA, "Pharmaceutical Sales Data," 2022.
- Research and Markets, "Emerging Markets in Allergy Drugs," 2023.