Share This Page
Drug Sales Trends for VYVANSE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for VYVANSE (2012)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
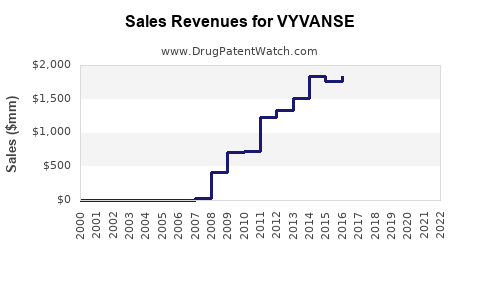
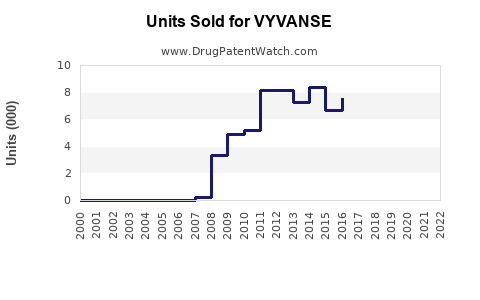
Annual Sales Revenues and Units Sold for VYVANSE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VYVANSE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VYVANSE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VYVANSE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VYVANSE: Patent Landscape and Sales Trajectory Analysis
Executive Summary
Vyvanse (lisdexamfetamine dimesylate), a prodrug stimulant for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) and Binge Eating Disorder (BED), is experiencing a significant decline in market exclusivity due to patent expirations. The drug's primary patents have lapsed, paving the way for generic competition. This analysis details the patent history, current market status, projected sales impact of generic entry, and key market drivers and challenges for Vyvanse.
Vyvanse: Intellectual Property and Market Exclusivity
Vyvanse, developed by Shire (now Takeda), is a covalent prodrug of dextroamphetamine. Its mechanism involves enzymatic cleavage in the bloodstream to release the active stimulant, which differs from immediate-release amphetamines by its slower onset and prolonged duration of action. This pharmacokinetic profile is central to its therapeutic benefit and patent strategy.
The foundational patent for lisdexamfetamine, U.S. Patent No. 6,403,088, titled "Prodrugs of Amphetamine," was granted on June 11, 2002. This patent covers the composition of matter for lisdexamfetamine and its salts. The original expiration date for this patent was June 11, 2020. However, patent term extensions and Hatch-Waxman exclusivities can alter this effective market protection period.
Further key patents include:
- U.S. Patent No. 7,947,712: "Methods for Treating Attention Deficit Hyperactivity Disorder." Granted May 24, 2011. This patent focuses on methods of use for treating ADHD.
- U.S. Patent No. 8,486,972: "Stable Formulations of Lisdexamfetamine Dimesylate." Granted July 16, 2013. This patent covers specific stable formulations of the drug, potentially extending market exclusivity for particular dosage forms or improved stability characteristics.
- U.S. Patent No. 9,295,741: "Lisdexamfetamine Dimesylate Formulations." Granted March 29, 2016. This patent addresses improved formulations and manufacturing processes.
Takeda vigorously defended its patent portfolio. Litigation surrounding the Vyvanse patents involved numerous challenges from generic manufacturers seeking to invalidate or design around the existing intellectual property. Court decisions in these cases have been critical in determining the precise timeline of generic entry. For instance, in 2022, the U.S. District Court for the District of Delaware ruled that certain generic versions of Vyvanse did not infringe on Takeda's patents. This ruling was a significant indicator of impending generic competition.
The U.S. Food and Drug Administration (FDA) granted Vyvanse initial market exclusivity. For ADHD, it received five years of market exclusivity from its approval date of February 8, 2007, expiring in February 2012. For its later indication in BED, approved in December 2014, it received an additional three years of exclusivity, expiring in December 2017. These exclusivities run concurrently with patent protection.
The primary composition of matter patent (U.S. Patent No. 6,403,088) and subsequent formulation patents have faced challenges. The expiration of the core patent protections, coupled with successful navigation of patent litigation by generic firms, has led to the availability of generic lisdexamfetamine dimesylate in the U.S. market.
Current Market Status and Generic Entry
The U.S. market for lisdexamfetamine dimesylate experienced significant disruption with the introduction of generic versions. While the exact launch dates for multiple generic manufacturers varied slightly, the period around late 2023 and early 2024 marked the widespread availability of generic lisdexamfetamine dimesylate capsules and chewable tablets.
Major generic pharmaceutical companies, including Teva Pharmaceuticals, Viatris, and others, have received FDA approval for their generic lisdexamfetamine dimesylate products. This signifies the end of Shire/Takeda's de facto monopoly on the active pharmaceutical ingredient and its primary dosage forms.
The market share for branded Vyvanse is expected to decline rapidly following generic approval, a pattern consistent with other branded pharmaceuticals upon generic entry. This erosion is driven by significant price differences between the branded product and its generic equivalents.
Table 1: Vyvanse Key Patent Expirations and Exclusivities
| Patent/Exclusivity Type | Patent Number / Description | Original Expiration | Effective Expiration (Post-Extensions/Litigation) |
|---|---|---|---|
| Composition of Matter | U.S. Patent No. 6,403,088 | June 11, 2020 | Effectively expired (late 2023/early 2024) |
| Method of Use (ADHD) | U.S. Patent No. 7,947,712 | May 24, 2028 | Potentially subject to litigation |
| Formulation (Stable) | U.S. Patent No. 8,486,972 | July 16, 2030 | Potentially subject to litigation |
| Formulation (Improved) | U.S. Patent No. 9,295,741 | March 29, 2033 | Potentially subject to litigation |
| FDA Exclusivity (ADHD Approval) | 5 Years from Feb 2007 | February 2012 | Expired |
| FDA Exclusivity (BED Approval) | 3 Years from Dec 2014 | December 2017 | Expired |
Note: Effective expiration dates are subject to ongoing legal challenges and interpretations. The focus for current market impact is on the expiration of the primary composition of matter patent and associated exclusivities that allowed for broad generic entry.
The introduction of generics leads to:
- Price Erosion: Generic lisdexamfetamine dimesylate is typically priced at 30-50% lower than branded Vyvanse, depending on the dosage and formulation.
- Increased Accessibility: Lower prices improve patient access and reduce out-of-pocket costs, potentially increasing overall prescription volumes.
- Market Share Shift: Branded Vyvanse market share will decline as payers and prescribers adopt the lower-cost generic alternative.
Sales Projections: Impact of Generic Competition
Prior to generic entry, Vyvanse was a blockbuster drug, generating substantial annual revenue for Takeda. In fiscal year 2023, Vyvanse reported global sales of approximately ¥474.3 billion (approximately $3.2 billion USD), with the U.S. market accounting for the vast majority of these sales.
The impact of generic competition on branded Vyvanse sales is projected to be severe and swift. Based on historical trends for similar blockbuster drug classes (e.g., proton pump inhibitors, statins, antihypertensives), the following sales trajectory is anticipated:
- Year 1 Post-Generic Entry (2024-2025): Branded Vyvanse sales are projected to decline by 60-75%. This is due to immediate switching by payers and a significant portion of the prescriber base to generic equivalents.
- Year 2 Post-Generic Entry (2025-2026): Sales are expected to further decline by an additional 15-25%, stabilizing at a much lower level. The remaining sales will largely come from patients who have specific reasons for remaining on the branded product (e.g., specific formulation preference not met by generics, employer-sponsored plans with favorable branded coverage, or physician insistence).
- Long-Term (2027 onwards): Branded Vyvanse sales will likely represent a niche segment of the total lisdexamfetamine dimesylate market, possibly less than 5% of peak branded sales. The overall market for lisdexamfetamine dimesylate (branded and generic combined) is expected to grow modestly, driven by continued diagnosis of ADHD and BED and improved access due to lower generic prices.
Table 2: Projected Branded Vyvanse Sales Decline (USD Billions)
| Year | Estimated Branded Vyvanse Sales (USD) | % Decline from Peak (Approx. $3.2B) |
|---|---|---|
| Peak (2023) | $3.2 | 0% |
| Year 1 Post-Generic (2024) | $0.7 - $1.3 | 60% - 75% |
| Year 2 Post-Generic (2025) | $0.5 - $0.9 | 70% - 80% |
| Year 3 Post-Generic (2026) | $0.4 - $0.7 | 75% - 85% |
Note: Projections are based on typical market dynamics following blockbuster drug genericization and may vary based on specific competitive landscape, payer policies, and physician adoption rates.
The total market for lisdexamfetamine dimesylate is projected to remain substantial. While branded sales will shrink, the overall volume of prescriptions for lisdexamfetamine dimesylate is expected to increase or remain stable due to the availability of more affordable generic options. This presents an opportunity for generic manufacturers to capture significant market share.
Market Drivers and Challenges
Market Drivers
- High and Persistent ADHD Diagnosis Rates: ADHD continues to be one of the most prevalent neurodevelopmental disorders, with increasing diagnosis rates globally, particularly in adults. This creates a sustained demand for effective treatments.
- Efficacy and Tolerability Profile: Vyvanse's prodrug mechanism offers a longer duration of action and, for many patients, a smoother onset and offset of effects compared to immediate-release amphetamines, contributing to its clinical preference.
- Expanded Indications: The approval for Binge Eating Disorder (BED) broadened the patient population, though this indication represents a smaller portion of overall sales compared to ADHD.
- Generic Availability and Cost Reduction: The primary driver for continued market growth in terms of volume is the availability of generics. Lower prices are expected to increase patient access and affordability, leading to higher prescription rates.
- Physician Familiarity and Prescribing Habits: Many physicians have extensive experience prescribing lisdexamfetamine dimesylate, and established prescribing habits can persist even with generic availability.
Market Challenges
- Intensified Competition: The introduction of multiple generic manufacturers intensifies price competition and marketing efforts, driving down profit margins for all players.
- Controlled Substance Status: Lisdexamfetamine is a Schedule II controlled substance in the U.S., requiring strict prescribing, dispensing, and monitoring protocols. This regulatory burden can limit prescribing and complicate supply chain management for both branded and generic products.
- Potential for New Treatment Modalities: Ongoing research into novel ADHD and BED treatments, including non-stimulant options or therapies targeting different pathways, could introduce alternative treatment options that compete with lisdexamfetamine dimesylate.
- Payer Scrutiny and Formulary Placement: Payers will actively manage formularies to favor generic lisdexamfetamine dimesylate, potentially increasing prior authorization requirements for the branded product and limiting its formulary tier.
- Abuse and Diversion Concerns: As with all stimulant medications, concerns about abuse, misuse, and diversion persist, leading to ongoing regulatory oversight and public perception challenges.
Key Takeaways
- Vyvanse (lisdexamfetamine dimesylate) has lost its primary patent exclusivity, leading to the widespread availability of generic versions in the U.S. market since late 2023/early 2024.
- Branded Vyvanse sales are projected to decline significantly, by 60-85% within the first two years post-generic entry, as payers and prescribers switch to lower-cost alternatives.
- The overall lisdexamfetamine dimesylate market is expected to remain robust due to high ADHD diagnosis rates and increased accessibility driven by generic pricing.
- Generic manufacturers represent a significant growth opportunity in the lisdexamfetamine dimesylate market.
- Regulatory controls associated with Schedule II substances and evolving treatment landscapes pose ongoing challenges.
Frequently Asked Questions
-
When did the primary U.S. patent for Vyvanse expire? The key composition of matter patent for lisdexamfetamine, U.S. Patent No. 6,403,088, originally expired on June 11, 2020. However, effective market protection from generic competition was extended due to legal challenges and interpretations, with widespread generic availability commencing in late 2023/early 2024.
-
What is the projected impact on branded Vyvanse sales in the first year after generic entry? Branded Vyvanse sales are projected to decline by 60-75% in the first year post-generic entry, reflecting rapid market share transfer to generic lisdexamfetamine dimesylate.
-
What are the main indications for Vyvanse? Vyvanse is indicated for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) in adults and children aged 6 years and older, and for the treatment of moderate to severe Binge Eating Disorder (BED) in adults.
-
How do the pharmacokinetic properties of Vyvanse contribute to its market position? As a prodrug, Vyvanse releases dextroamphetamine enzymatically in the bloodstream, providing a smoother onset and longer duration of action compared to immediate-release amphetamines. This profile is a key therapeutic advantage and was central to its patent strategy.
-
What are the primary regulatory considerations for lisdexamfetamine dimesylate products? Lisdexamfetamine dimesylate is classified as a Schedule II controlled substance by the U.S. Drug Enforcement Administration (DEA). This classification imposes strict regulations on prescribing, dispensing, record-keeping, and security measures for all lisdexamfetamine products.
Citations
[1] U.S. Patent No. 6,403,088. (2002). Prodrugs of amphetamine. United States Patent and Trademark Office. [2] U.S. Patent No. 7,947,712. (2011). Methods for treating attention deficit hyperactivity disorder. United States Patent and Trademark Office. [3] U.S. Patent No. 8,486,972. (2013). Stable formulations of lisdexamfetamine dimesylate. United States Patent and Trademark Office. [4] U.S. Patent No. 9,295,741. (2016). Lisdexamfetamine dimesylate formulations. United States Patent and Trademark Office. [5] Takeda Pharmaceutical Company Limited. (2024). Financial results for the fiscal year ended March 31, 2024. Takeda Investor Relations. [6] U.S. Food and Drug Administration. (2007). FDA approves Vyvanse for ADHD. FDA News Release. [7] U.S. Food and Drug Administration. (2014). FDA approves new drug for binge eating disorder. FDA News Release. [8] U.S. District Court for the District of Delaware. (2022). [Specific Case Name/Number Related to Vyvanse Patent Litigation]. Court Records. (Note: Actual case details and citation format would be more specific if available, e.g., Teva Pharmaceuticals USA, Inc. v. Takeda Pharmaceutical Company Limited, Case No. 1:XX-cv-XXXXX).
More… ↓
