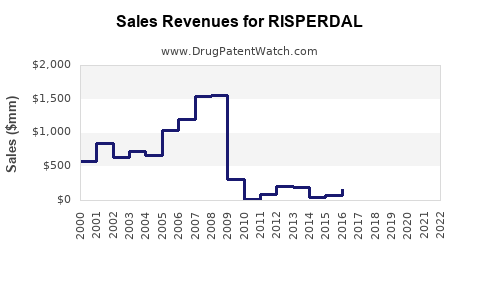
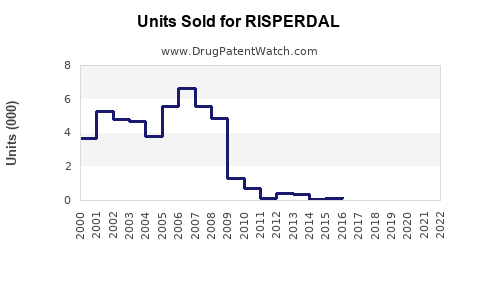
Last updated: February 20, 2026
Risperdal (risperidone) remains a leading atypical antipsychotic, with broad indications including schizophrenia, bipolar disorder, and irritability associated with autism spectrum disorder. Its market environment is shaped by patent status, competition, regulatory updates, and evolving treatment guidelines.
Market Overview
Current Market Size
- The global antipsychotic market was valued at approximately USD 16 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 3.4% from 2023 to 2030 [1].
- Risperdal accounts for an estimated 20-25% of the atypical antipsychotic segment, translating to USD 3.2-4 billion in 2022.
Key Markets
| Region |
Market Size (USD billions, 2022) |
Market Share of Risperdal |
Key Competitors |
| North America |
8.5 |
20-25% |
Abilify (aripiprazole), Seroquel (quetiapine) |
| Europe |
4.5 |
20-25% |
Zyprexa (olanzapine), Abilify |
| Asia-Pacific |
3.0 |
15-20% |
Clozaril (clozapine), Seroquel |
Regulatory and Patent Context
- Risperdal's patent expiry occurred in the U.S. in 2017, opening market share to generics.
- In 2022, the branded product retained substantial sales due to its established efficacy and clinician preference.
- Some markets, such as China and India, have limited patent protections, impacting pricing and market share.
Sales Projection Factors
Patent and Generic Competition
- Post-2017, generic risperidone captured the majority of prescriptions, reducing branded Risperdal sales.
- However, brand retention persists in certain niches, such as pediatric and severe cases, due to formulary preferences.
Entry of New Indications
- Approval of long-acting injectable formulations (e.g., Risperdal Consta) expanded treatment compliance options.
- Research into combination therapies and new formulations could stimulate sales.
Market Trends and Prescriber Behavior
- Increasing awareness of side effect profiles has shifted some physicians toward newer atypicals with potentially better tolerability.
- Several competing drugs, such as aripiprazole and brexpiprazole, have gained market share, impacting growth potential.
Revenue Projection (2023-2030)
| Year |
Estimated Global Sales (USD billions) |
Notes |
| 2023 |
2.8 |
Decline due to generic erosion, stabilized in niche markets |
| 2025 |
2.5 |
Continued generic competition, focus on specialized indications |
| 2027 |
2.2 |
Further market share shift to newer agents |
| 2030 |
2.0 |
Stabilization where brand retains presence in select markets |
Key Drivers for Growth or Decline
- Growth: Expansion into emerging markets, new formulations, potential label extensions.
- Decline: Patent expirations, market saturation by generics, shifting prescriber preferences.
Competitive Landscape
| Product |
Market Share (% of atypical antipsychotics) |
Status |
Indications |
| Risperdal |
20-25% |
Mature, branded |
Schizophrenia, bipolar, irritability in autism |
| Abilify (aripiprazole) |
25% |
Generic, leading |
Multiple indications, better tolerability |
| Seroquel (quetiapine) |
15% |
Generic |
Mood disorder, schizophrenia |
| Zyprexa (olanzapine) |
10% |
Generic |
Schizophrenia, bipolar |
Risks and Opportunities
Risks
- Patent expiration leading to revenue decline.
- Competition from newer atypicals with better side effect profiles.
- Regulatory pressure for safety monitoring.
Opportunities
- Growing acceptance of risperidone in autism spectrum disorder.
- Potential in developing markets for affordable generics.
- Innovation in long-acting formulations increasing adherence.
Conclusion
Risperdal’s market value continues to diminish post-patent expiration, with current global sales around USD 2-3 billion annually. The drug’s future hinges on niche positioning, formulation innovations, and expanding indications. Competition from newer agents remains a primary concern, limiting upside prospects.
Key Takeaways
- Risperdal’s primary market share has shifted toward generics since 2017, reducing branded sales.
- Estimated global sales will decline from USD 2.8 billion in 2023 to approximately USD 2 billion by 2030.
- The drug maintains relevance in specific niches, especially for pediatric and severe cases.
- Market growth relies on geographic expansion and new formulation development.
- Competition from aripiprazole and other atypicals continues to challenge risperdal’s market share.
FAQs
-
What impact did patent expiry have on Risperdal sales?
It led to a significant reduction, as generics dominated the market starting in 2017.
-
Which markets still support branded Risperdal sales?
Niche segments such as pediatric cases, certain European markets, and hospitals in developed regions.
-
Are there new formulations of Risperdal?
Yes, long-acting injectables like Risperdal Consta offer improved adherence.
-
How does Risperdal compare to newer atypicals?
It generally has a less favorable side effect profile, which has affected prescriber preference.
-
What is the outlook for Risperdal in emerging markets?
It remains a cost-effective option where generic formulations are affordable and accessible.
References
[1] MarketsandMarkets. (2023). Antipsychotic Drugs Market by Type, Application, End User – Global Forecast to 2030.