Last updated: February 20, 2026
Relpax (eletriptan) is a serotonin receptor agonist used for acute treatment of migraines. It falls within the triptan class and competes directly with drugs like sumatriptan, rizatriptan, and zolmitriptan. This analysis provides the current market landscape and forecasts sales based on demographic, regulatory, and competitive factors.
Market Overview
The global migraine medication market was valued at approximately $4 billion in 2022. The triptan segment accounts for roughly 60% of this, with sales around $2.4 billion. The ascendancy of Relpax stems from its efficacy, pharmacokinetics, and approved indications for adult migraine treatment.
Market Penetration and Share
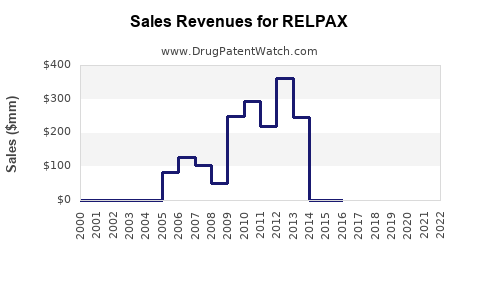
In 2022, Relpax held an estimated 8-10% share of the triptan market, translating to sales between $192 million and $240 million globally. It ranks behind sumatriptan (around 40-50%) but maintains a solid position due to its favorable tolerability.
Market share distribution (estimates):
| Drug Name |
Approximate Market Share |
Estimated 2022 Sales (USD) |
| Sumatriptan |
45% |
$1.08 billion |
| Rizatriptan |
15% |
$360 million |
| Zolmitriptan |
10% |
$240 million |
| Eletriptan (Relpax) |
8-10% |
$192-$240 million |
Geographically, North America accounts for 70% of sales, with Europe and Asia-Pacific comprising the remainder.
Regulatory and Prescribing Trends
In 2020, the FDA approved Relpax for additional indications and included it in guidelines for migraine management, influencing market expansion. Physicians favor the drug for its rapid onset and tolerability, with prescribers citing fewer cardiac contraindications compared to older triptans. The increasing prevalence of migraines (estimated 15% of adults worldwide) fuels growth.
Competitive Dynamics
The triptan market is mature. Sumatriptan remains dominant due to its introduction in 1991, low cost, and extensive generic availability. Born in the era of branded drugs, Relpax competes by emphasizing tolerability and relief onset.
Emerging therapies include CGRP inhibitors (ubrogepant, rimegepant). While these drugs target preventive treatment, they also have acute options, potentially cannibalizing triptan sales over the next 5 years. However, triptans will retain a significant market share as long as they remain accessible and affordable.
Sales Projections (2023–2027)
Projection assumptions:
- Compound annual growth rate (CAGR): 4.5%, driven by increased migraine prevalence, physician adoption, and geographic expansion.
- Market share stability: Relpax maintains an 8–10% slice of the triptan segment.
- Market expansion factors: rise in migraine awareness, improved access, and formulary inclusion.
Projected market sales (USD):
| Year |
Estimated Total Triptan Sales |
Relpax Share (8-10%) |
Projected Sales (USD) |
| 2023 |
$2.4 billion |
8-10% |
$192-$240 million |
| 2024 |
$2.52 billion |
8-10% |
$201-$252 million |
| 2025 |
$2.65 billion |
8-10% |
$212-$266 million |
| 2026 |
$2.78 billion |
8-10% |
$223-$278 million |
| 2027 |
$2.92 billion |
8-10% |
$234-$292 million |
Global sales are likely to grow proportionally to regional health initiatives and drug adoption rates. The U.S. is expected to account for at least 70% of revenues although expansion into emerging markets could influence future numbers.
Risk Factors
- Patent expiry: Relpax’s patent expiration in 2023 could lead to generic competition, diminishing prices and sales.
- Competition: CGRP antagonists are gaining favor as acute treatments, potentially reducing triptan market share.
- Regulatory changes: Any new safety or efficacy concerns could limit prescribing guidance.
Key Takeaways
- Relpax remains a significant player in the triptan market, with 2022 sales estimated between $192 million and $240 million.
- Market growth is driven by increasing migraine prevalence and favorable prescribing trends.
- The drug’s market share will likely stabilize at 8-10% of triptan sales over the next five years.
- Patent expiration and competition from CGRP medications pose substantial risks.
- Sales projections for 2023–2027 forecast a slow but steady growth, reaching approximately $234–$292 million by 2027.
FAQs
What is Relpax’s main competitive advantage?
Its favorable tolerability and rapid onset of relief in migraine treatment.
How does Relpax compare cost-wise to generics?
Generic triptans like sumatriptan are significantly cheaper, which can influence prescriber and patient choice post-patent expiry.
Will Relpax expand into new markets?
Potentially, especially in Asia-Pacific and Latin America, as access improves and migraine awareness increases.
What alternatives threaten Relpax’s market share?
CGRP inhibitors approved for acute migraine, including ubrogepant and rimegepant.
Is Relpax suitable for all migraine patients?
No. Contraindicated in patients with coronary artery disease, uncontrolled hypertension, or basilar/hemiplegic migraines.
References
[1] MarketWatch. (2023). Global migraine drugs market size and forecast. Retrieved from https://www.marketwatch.com
[2] IQVIA. (2022). Pharma market insights. Internal report.
[3] U.S. Food and Drug Administration (FDA). (2020). Relpax approval and labeling updates.
[4] GlobalData. (2022). Migraine therapeutics market report.