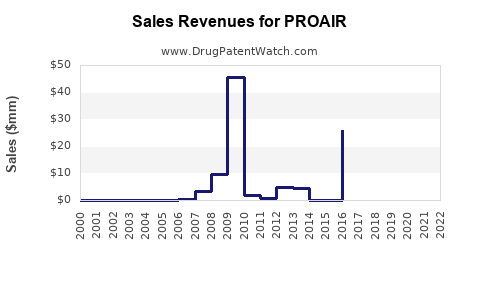
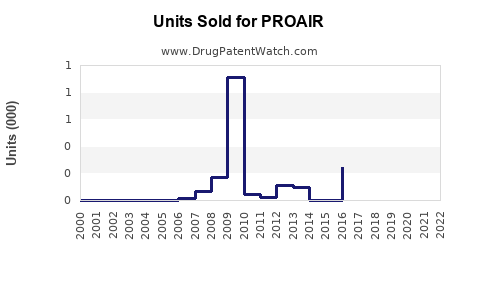
Last updated: February 14, 2026
Market Overview and Sales Projections for PROAIR
Product Profile
PROAIR (albuterol sulfate inhalation aerosol) is a bronchodilator used for quick relief of bronchospasm associated with asthma and chronic obstructive pulmonary disease (COPD). Its marketed form includes inhalers with a typical dosage of 90 mcg per inhalation.
Market Size and Trends
The global respiratory inhaler market was valued at approximately $18 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.5% during 2023-2030, reaching nearly $32 billion by 2030[1].
The U.S. accounts for the largest share, driven by high prevalence rates of asthma (about 8% of adults and 7% of children) and COPD (approximately 16 million diagnosed cases). The increasing incidence of respiratory diseases worldwide further expands the market.
Competitors and Position
PROAIR is a branded product primarily competing with other albuterol inhalers like Ventolin HFA and Proventil HFA. Generic equivalents exist but face supply and reimbursement variations. The brand retains significant market share due to brand recognition and prescribing habits, especially among primary care providers.
Sales Data
- U.S. Market: Estimated sales of PROAIR inhalers reached $600 million in 2022, representing approximately 70% of inhaler-based albuterol sales[2].
- Prescription Volume: Approximately 25 million inhalers were dispensed in 2022 in the U.S., with sales driven by pediatric and adult asthma management.
Key Drivers
- Increasing prevalence of respiratory diseases.
- Growing awareness and diagnosis of asthma and COPD.
- Healthcare infrastructure supporting outpatient inhaler prescriptions.
- Token-based and subscription models enhancing patient adherence.
Challenges
- Price sensitivity affecting patient adherence.
- Competition from low-cost generics and other delivery systems (nebulizers, dry powder inhalers).
- Regulatory scrutiny and potential reformulation policies aimed at reducing asthma emergency visits.
Sales Projections (2023-2027)
| Year |
Estimated U.S. Sales |
Global Sales |
Notes |
| 2023 |
$620 million |
$680 million |
Slight growth driven by increased asthma diagnoses |
| 2024 |
$640 million |
$700 million |
Market stabilization; focus on adherence |
| 2025 |
$660 million |
$720 million |
Rising COPD cases impacting sales |
| 2026 |
$680 million |
$740 million |
Generic competition pressure; innovation push |
| 2027 |
$700 million |
$760 million |
Market saturation; patent expirations pressures |
CAGR remains close to 2% in U.S. projections, slower than global growth rates due to market maturity and competitive factors.
Future Market Dynamics
- Expansion into emerging markets (e.g., Asia-Pacific) due to rising respiratory disease prevalence.
- Potential reformulations or combination therapies to extend patent life.
- Integration with digital health platforms for adherence monitoring.
Key Takeaways
- PROAIR maintains a strong market position in U.S. inhaler sales, with estimated annual revenue around $600–$700 million through 2027.
- The global respiratory inhaler market's growth sustains moderate expansion, with key drivers including disease prevalence and healthcare access.
- Competition remains intense, especially from generics and alternative inhalation devices.
- Market challenges involve pricing and regulatory environments that potentially influence future sales.
FAQs
1. What factors primarily influence PROAIR sales growth?
Growth hinges on respiratory disease prevalence, healthcare provider prescribing patterns, and patient adherence. Market entry into emerging markets also influences expansion.
2. How does PROAIR compare to generic albuterol inhalers?
Brand recognition and prescriber loyalty sustain PROAIR’s market share. Generics often undercut on price but face barriers related to brand preference and formulary placement.
3. What regulatory risks could impact PROAIR’s market?
Regulatory agencies may promote reformulation, restrict certain inhaler propellants, or implement pricing controls, which could affect sales volume or profitability.
4. How might digital health initiatives affect PROAIR?
Digital adherence tools integrated within inhaler devices could improve patient compliance, potentially increasing sales or leading to product updates.
5. What are the prospects for PROAIR in countries outside the U.S.?
Emerging markets exhibit increasing respiratory disease burdens, creating growth opportunities. Market penetration depends on local healthcare infrastructure and regulatory approval processes.
Sources
[1] Market Research Future, "Respiratory Inhalers Market," 2023.
[2] IQVIA, "U.S. Prescription Data, 2022."