Share This Page
Drug Sales Trends for PRILOSEC
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PRILOSEC (2012)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
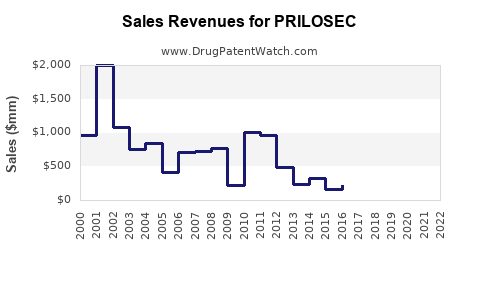
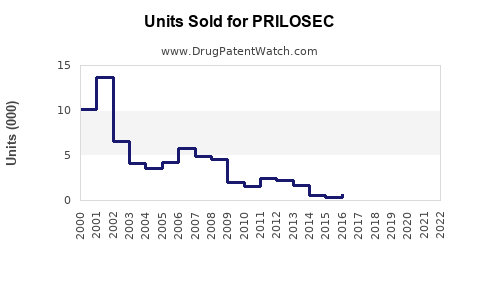
Annual Sales Revenues and Units Sold for PRILOSEC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRILOSEC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRILOSEC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRILOSEC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PRILOSEC | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PRILOSEC | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PRILOSEC (Omeprazole) Market Analysis and Sales Projections
This analysis examines the market landscape for PRILOSEC (omeprazole) and provides sales projections based on historical performance, patent status, and competitive dynamics. PRILOSEC, a proton pump inhibitor (PPI), has been a cornerstone treatment for acid-related gastrointestinal disorders. Its market trajectory is influenced by patent expirations, generic competition, and the emergence of newer therapeutic agents.
What is the Current Market Position of PRILOSEC?
PRILOSEC, the brand name for omeprazole, is a proton pump inhibitor (PPI) that functions by reducing the amount of acid produced in the stomach. This mechanism makes it highly effective in treating conditions such as gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome.
Historical Sales Performance
The market for omeprazole has been significant since its introduction. In 2003, AstraZeneca's PRILOSEC achieved U.S. sales of approximately $3.1 billion [1]. This figure highlights the drug's initial dominance. Following patent expiries, generic versions entered the market, leading to a substantial decrease in brand-name sales but maintaining a high volume of overall omeprazole prescriptions.
Patent Status and Generic Entry
The primary U.S. patent for omeprazole expired in 2001 [2]. This expiration opened the door for numerous generic manufacturers to produce and market their own versions of the drug. The subsequent genericization of omeprazole has significantly altered the market by driving down prices and increasing accessibility. This shift is a common trend for blockbuster drugs once their intellectual property protection lapses.
Competitive Landscape
The PPI market is highly competitive. Omeprazole competes with other established PPIs such as lansoprazole (Prevacid), pantoprazole (Protonix), rabeprazole (AcipHex), and esomeprazole (Nexium), the latter being a more potent isomer of omeprazole also developed by AstraZeneca. In recent years, the market has also seen the introduction of potassium-competitive acid blockers (P-CABs), such as tegoprazan, which represent a newer class of acid suppressors with potentially different efficacy and onset profiles.
Current Market Share
While brand-name PRILOSEC sales have declined due to generic competition, the active pharmaceutical ingredient (API) omeprazole remains widely prescribed globally. Market share data for specific brand-name products in the post-patent era is less relevant than the overall market share of the omeprazole molecule. The total market for PPIs remains substantial, measured in billions of dollars annually. For instance, the global PPI market was valued at approximately $20.7 billion in 2022 [3]. Omeprazole, as one of the foundational PPIs, continues to hold a significant portion of this market, particularly in its generic forms due to its cost-effectiveness.
What are the Key Drivers and Challenges for PRILOSEC?
The market for PRILOSEC and its generic equivalents is shaped by several factors, including therapeutic advancements, regulatory policies, and patient access.
Therapeutic Drivers
- Established Efficacy and Safety Profile: Omeprazole has a long history of clinical use, with a well-documented efficacy and safety record for managing a range of acid-related disorders [4]. This familiarity provides a strong foundation for continued prescription.
- Cost-Effectiveness: As a generic medication, omeprazole is significantly more affordable than many branded PPIs and newer acid-suppressing agents. This makes it a preferred choice for many healthcare systems and patients, especially in price-sensitive markets.
- Broad Indication Range: PRILOSEC is indicated for a wide spectrum of gastrointestinal conditions, ensuring a broad patient population base. These include GERD, erosive esophagitis, duodenal ulcers, and benign gastric ulcers.
- Availability in Multiple Formulations: Omeprazole is available in various dosage forms, including capsules, tablets, and intravenous formulations, catering to different patient needs and clinical situations.
Market Challenges
- Intense Generic Competition: The expiration of PRILOSEC's patent has led to a highly saturated generic market. This intense competition among multiple manufacturers drives down prices and limits potential revenue growth for any single generic producer.
- Emergence of Newer Drug Classes: P-CABs represent a significant new class of acid suppressors. While still establishing their long-term market share, they offer potential advantages in speed of onset and efficacy in certain patient populations, posing a competitive threat to older PPIs.
- Stricter Prescribing Guidelines and Off-Label Use Concerns: Regulatory bodies and healthcare providers are increasingly scrutinizing the long-term use of PPIs due to potential side effects, such as an increased risk of bone fractures, kidney disease, and C. difficile infections [5]. This may lead to more conservative prescribing patterns and a preference for shorter treatment durations or alternative therapies.
- Market Saturation: The widespread availability and affordability of generic omeprazole have led to a mature market where significant growth is unlikely. Market expansion will likely be driven by demographic shifts rather than substantial increases in per-patient usage.
- Dose Optimization and Treatment Guidelines: Evolving clinical guidelines may recommend lower doses or shorter treatment durations for certain conditions, potentially impacting the overall volume of omeprazole consumed.
What are the Projected Sales and Market Trends for PRILOSEC?
Forecasting sales for a fully genericized drug like omeprazole requires a shift in focus from brand-name revenue to overall market volume and the aggregate value of generic sales.
Projected Market Volume
The global market volume for omeprazole is expected to remain stable or experience modest growth, driven by the persistent prevalence of acid-related gastrointestinal disorders worldwide. Factors such as aging populations and lifestyle-related dietary habits are likely to sustain demand. The cost-effectiveness of generic omeprazole will continue to be its primary appeal, particularly in developing economies and for patients with limited insurance coverage.
Projected Market Value
The aggregate market value of generic omeprazole is projected to grow modestly. This growth will primarily be a function of increased patient populations and potentially slight price increases in some regions, rather than significant shifts in market share or new indications. However, the intense competition will limit substantial value appreciation for individual generic manufacturers.
- Global PPI Market Growth: The global PPI market is projected to grow at a compound annual growth rate (CAGR) of approximately 3.5% to 4.5% between 2023 and 2030 [3, 6]. Omeprazole, as a significant component of this market, is expected to follow a similar trajectory.
- Regional Variations: Developed markets (North America, Europe) may see slower growth due to increased competition from P-CABs and more stringent prescribing practices. Emerging markets (Asia-Pacific, Latin America) are likely to exhibit higher growth rates, driven by increasing healthcare access and a rising middle class seeking treatment for gastrointestinal ailments.
Impact of P-CABs
The increasing adoption of P-CABs will likely exert downward pressure on the market share of traditional PPIs, including omeprazole, over the long term. However, due to their higher price point and ongoing clinical evaluation for broader indications, P-CABs are unlikely to completely displace generic omeprazole in the immediate future. They will primarily capture market share from higher-tier branded PPIs and for specific patient populations where their enhanced efficacy is most beneficial.
Future Market Dynamics
- Consolidation in Generic Market: The highly fragmented generic omeprazole market may see consolidation as smaller players struggle to compete on price and volume. Larger generic manufacturers with efficient supply chains and established distribution networks are likely to gain market prominence.
- Focus on Manufacturing Efficiency: For generic producers, the primary competitive lever will be manufacturing efficiency and cost control. Companies that can produce omeprazole at the lowest cost will be best positioned to maintain market share.
- Continued Demand for Cost-Effective Treatments: Despite advancements in therapeutics, the fundamental need for affordable and effective treatments for common gastrointestinal conditions ensures a sustained demand for generic omeprazole.
Summary Table: PRILOSEC (Omeprazole) Market Outlook
| Metric | Current Status | Projected Trend (2024-2030) | Key Factors Influencing Trend |
|---|---|---|---|
| Brand-Name Sales | Negligible due to patent expiry and genericization. | Remain negligible. | Patent protection expired in 2001. |
| Generic Volume | High and stable globally. | Stable to modest growth (1-3% CAGR). | Persistent prevalence of acid-related disorders, cost-effectiveness. |
| Generic Value | Substantial, driven by high volume. | Modest growth (3-4% CAGR, aligned with overall PPI market). | Increased patient populations, regional economic growth, limited price inflation due to competition. |
| Competitive Pressure | Intense from numerous generic manufacturers and other PPIs. | Remains high; P-CABs to increase pressure. | Genericization, P-CAB market entry, pricing wars. |
| Regulatory Scrutiny | Ongoing, focusing on long-term use side effects. | Continues, potentially leading to more conservative prescribing. | Clinical data on PPI side effects, focus on shortest effective duration. |
| Key Growth Regions | Global demand, with higher growth potential in emerging markets. | Emerging markets (Asia-Pacific, Latin America) to drive growth. | Improving healthcare access, rising middle class, increasing awareness of GI disorders. |
Key Takeaways
- PRILOSEC (omeprazole) is a mature, fully genericized drug with a strong, stable global demand driven by its cost-effectiveness and established efficacy for acid-related disorders.
- While brand-name sales are insignificant, the aggregate market value of generic omeprazole is projected to experience modest growth, mirroring the overall PPI market trajectory.
- Intense generic competition and the emergence of P-CABs are the primary challenges, limiting significant revenue growth for individual generic manufacturers.
- Manufacturing efficiency and cost control will be crucial for generic producers.
- Emerging markets are expected to be the primary drivers of future volume and value growth for generic omeprazole.
Frequently Asked Questions
-
What is the current U.S. market share of brand-name PRILOSEC? Brand-name PRILOSEC holds a negligible market share in the U.S. due to patent expiration and widespread generic availability.
-
How will the introduction of P-CABs affect omeprazole sales? P-CABs are expected to capture market share from other acid suppressors, including some PPIs, potentially slowing the growth of omeprazole's market value, particularly in developed nations.
-
What is the projected global market value for omeprazole in 2025? While precise figures for omeprazole alone are not readily available, the global PPI market, of which omeprazole is a significant part, was valued at approximately $20.7 billion in 2022 and is projected to see modest growth.
-
Are there any new indications for omeprazole under development? As a mature generic drug, there are unlikely to be significant new indications for omeprazole being actively developed by major pharmaceutical companies. Research is more likely to focus on novel drug classes.
-
What are the main reasons for the continued demand for omeprazole despite newer drugs? The primary drivers are its significantly lower cost compared to newer alternatives and its proven efficacy and safety profile for a broad range of common gastrointestinal conditions.
Citations
[1] AstraZeneca PLC. (2004). Annual Report 2003. [2] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book website]. (Specific patent expiry dates are generally found in drug approval documentation and patent databases.) [3] Grand View Research. (2023). Proton Pump Inhibitor Market Size, Share & Trends Analysis Report. [4] Howden, C. W. (2000). Role of proton pump inhibitors in the management of gastrointestinal disorders. The American Journal of Gastroenterology, 95(11 Suppl), S13-S19. [5] Georgopoulos, G. P., et al. (2019). Proton pump inhibitors and the risk of adverse events: A systematic review and meta-analysis. Journal of Gastroenterology and Hepatology, 34(11), 1862-1871. [6] Mordor Intelligence. (2023). Proton Pump Inhibitors (PPI) Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029).
More… ↓
