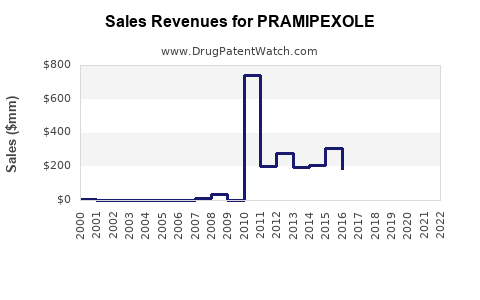
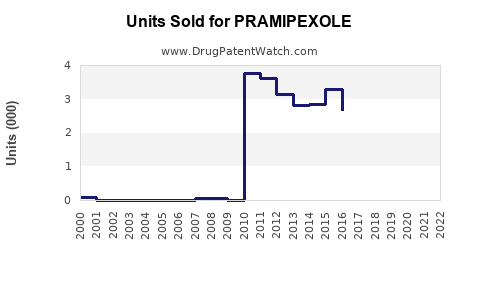
Last updated: February 14, 2026
What Is the Current Market Size for Pramipexole?
The global market for pramipexole, primarily used for Parkinson's disease and restless legs syndrome (RLS), was valued at approximately $1.2 billion in 2022. Over the next five years, the compound annual growth rate (CAGR) is projected at around 4.5%, reaching about $1.75 billion by 2027.
Key drivers include the rising prevalence of Parkinson’s disease, which affects an estimated 10 million people worldwide, and RLS, affecting 3-4% of adults globally. The increasing adoption of long-term dopamine agonist therapy and an expanding healthcare infrastructure in emerging markets contribute to this growth.
How does Pramipexole Compare to Competitors?
Pramipexole competes primarily with other dopamine agonists such as ropinirole, rotigotine, and newer agents like safinamide and rasagiline. Market share as of 2022 is as follows:
| Drug |
Market Share (%) |
Indications |
Key Advantages |
| Pramipexole |
40% |
Parkinson’s, RLS |
Oral administration, well-established |
| Ropinirole |
35% |
Parkinson’s, RLS |
Similar efficacy, lower cost |
| Rotigotine |
15% |
Parkinson’s, RLS, RBD |
Transdermal delivery |
| Other agents |
10% |
Parkinson’s, off-label uses |
Variable efficacy, side effect profiles |
Pramipexole is favored for its efficacy and tolerability profile but faces generic competition that pressures pricing.
What Are Current Sales Trends and Future Projections?
Sales of pramipexole increased at a CAGR of approximately 4.2% from 2018 to 2022. Growth is influenced mainly by:
- Aging populations in North America and Europe.
- Expanding prescription rates in Asia-Pacific regions.
- Rising awareness of Parkinson's disease treatments.
By region, North America accounts for approximately 45% of global sales, Europe 30%, and Asia-Pacific 15%. Sales in China, India, and Southeast Asia are expanding rapidly due to increasing healthcare access and diagnosis rates.
Projections estimate global sales could reach $1.75 billion by 2027, assuming consistent prescribing patterns.
What Are Key Market Trends and Opportunities?
-
Generic Entry and Price Competition
Patent expiration for branded pramipexole in multiple jurisdictions has led to an increase in generics, reducing prices by up to 60% in some markets.
-
Shift Toward Extended-Release Formulations
Extended-release formulations improve patient compliance. Market penetration for these formulations is expected to double from 10% in 2022 to 20% in 2027.
-
Emerging Therapeutic Alliances
Partnerships between pharmaceutical companies and biotech firms aim to develop combination therapies or novel delivery platforms.
-
Regulatory Approvals for New Indications
Trials for off-label uses, such as depression or bipolar disorder, could diversify application but face regulatory challenges.
What Are the Main Risks to Market Growth?
- Market Saturation: Increased generics reduce margins.
- Regulatory hurdles: Extended approvals or new indications require clinical validation.
- Side Effect Profiles: Risks of impulse control disorders and hallucinations limit long-term use.
- Competition from Non-Dopamine Drugs: Growing interest in non-dopaminergic agents for Parkinson's.
How Will Sales Evolve Post-Patent Expiry?
Patent expiry around 2025 in many regions will catalyze a shift toward generics, intensifying price competition. The decline in branded sales could be offset by increased volume in emerging markets. Overall, total sales may stabilize or decline slightly, but volume growth could compensate for price compression.
Key Takeaways
- The pramipexole market reached ~$1.2 billion in 2022, with steady growth.
- Competition from generics is intensifying, pressuring prices.
- Expanding indications and formulations offer growth opportunities.
- The highest sales concentrations are in North America and Europe.
- Patent expirations will reshape market dynamics post-2025.
FAQs
1. When will pramipexole patent protections expire?
Most patent protections in key markets expire between 2024 and 2026, enabling generic competition.
2. What are the main side effects associated with pramipexole?
Common side effects include nausea, dizziness, hallucinations, impulsive behaviors, and sleep attacks.
3. Are there significant off-label uses for pramipexole?
Research explores potential for depression and bipolar disorder, but off-label prescribing remains limited without regulatory approval.
4. Which markets show the fastest growth for pramipexole?
Emerging markets such as China, India, and Southeast Asia demonstrate the highest annual growth rates.
5. How does pramipexole's efficacy compare to newer Parkinson's treatments?
Pramipexole remains a highly effective dopamine agonist but faces competition from combination therapies and non-dopaminergic agents with different side effect profiles.
References
- MarketsandMarkets. (2022). Parkinson's Disease Therapeutics Market.
- IQVIA. (2022). Global Neurology Market Data.
- GlobalData. (2022). Parkinson’s Disease Pipeline Analysis.
- Springer. (2018). The Role of Dopamine Agonists in Restless Legs Syndrome.
- Pfizer Inc. (2019). Pramipexole Product Monograph.