Last updated: February 14, 2026
Overview
NOVOLIN 70/30, a premixed insulin consisting of 70% insulin lispro protamine and 30% insulin lispro, targets type 2 and type 1 diabetes management. Its rapid-acting component provides meal-time control, while the protamine component offers basal coverage. Positioned in the insulin market, it faces competition from similar premixed insulins like Humalog Mix 75/25, NovoLog Mix 70/30, and generics. The insulin market exhibits steady growth driven by increasing diabetes prevalence and adoption of insulin therapies.
Market Size and Trends
- The global insulin market was valued at approximately $25 billion in 2022.
- It is projected to grow at a compound annual growth rate (CAGR) of 6-8% through 2030, reaching an estimated $45 billion.
- The segment of premixed insulins accounts for roughly 40% of total insulin sales.
- Growth drivers include rising diabetes incidence, especially in Asia-Pacific, and increased adoption of insulin analogs over human insulins.
Regulatory Status
- NOVOLIN 70/30 is approved by the U.S. Food and Drug Administration (FDA) and other major regulatory bodies.
- The approval process emphasizes safety, efficacy, and manufacturing standards consistent with insulin analogs.
Competitive Position
| Insulin Product |
Market Share (Estimated, 2022) |
Price Range (per unit) |
Notable Features |
| NOVOLIN 70/30 |
15-20% |
$0.20 - $0.30 |
Premixed, fast-acting component derived from insulin lispro. |
| Humalog Mix 75/25 |
20-25% |
$0.25 - $0.35 |
Similar premix with slightly different formulation. |
| NovoLog Mix 70/30 |
25-30% |
$0.22 - $0.32 |
Preferred in certain regions; similar pharmacokinetics. |
| Generics and biosimilars |
10-15% |
$0.15 - $0.25 |
Entering phase; price pressures may increase. |
Sales Projections
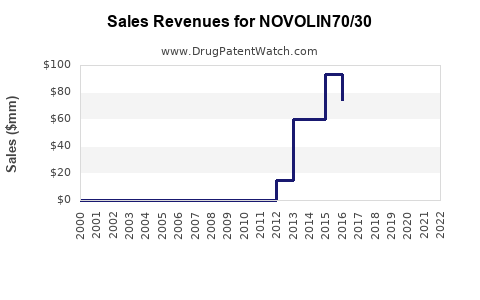
- 2023: Estimated sales of NOVOLIN 70/30 approximately $400–$500 million worldwide.
- 2025: Targeted growth anticipates a 10-15% increase year-over-year, reaching $600–$750 million as adoption expands, especially in emerging markets.
- 2030: Cumulative sales could surpass $1 billion, factoring in increased insulin use and a shift toward insulin analogs among patients with poorly controlled diabetes.
Key Factors Influencing Sales Growth
- Market Penetration: Limited to regions where Novo Nordisk markets NOVOLIN, primarily North America, Europe, and select emerging markets.
- Pricing Strategies: Competitive pricing will be critical against biosimilars and newer insulin formulations.
- Physician and Patient Acceptance: Preference for flexible, reliable insulin regimens impacts prescription volume.
- Regulatory Approvals: Approval of biosimilars may affect market share and pricing dynamics.
- Diabetes Prevalence Growth: Rising in regions like Asia-Pacific, Latin America, fueled by lifestyle changes.
Distribution Channels
- Insurers and pharmacy benefit managers dominate distribution.
- Hospitals and clinics account for a significant portion, especially in hospital-based care.
- Increasing online pharmacy sales, particularly post-2020 regulatory adjustments.
Challenges
- Patent expirations in the insulin market could lead to increased biosimilar competition.
- Reimbursement policies vary across regions, influencing accessibility.
- Patient adherence remains a concern due to injection frequency and treatment complexity.
Conclusion
NOVOLIN 70/30 positions as a mid-tier insulin product with steady growth prospects. Its success hinges on market penetration, pricing strategies, and the evolving competitive landscape. Accelerated growth scenarios depend on expanding into emerging markets and maintaining regulatory approval pathways.
Key Takeaways
- The global insulin market is projected to grow at 6-8% annually, reaching $45 billion by 2030.
- Premixed insulins like NOVOLIN 70/30 account for about 40% of the market with steady sales growth.
- Sales are expected to reach $600–$750 million by 2025 and potentially exceed $1 billion by 2030.
- Competitive pressure from biosimilars and generics could impact future market share.
- Pricing, reimbursement, and physician acceptance are critical factors for growth.
FAQs
-
How does NOVOLIN 70/30 compare to other premixed insulins in terms of efficacy?
It provides similar glycemic control compared to competitors like Humalog Mix 75/25 and NovoLog Mix 70/30, with comparable pharmacokinetics.
-
What markets offer the greatest growth potential for NOVOLIN 70/30?
Emerging markets in Asia-Pacific and Latin America show high potential due to rising diabetes prevalence and expanding healthcare infrastructure.
-
Will biosimilars threaten NOVOLIN 70/30 sales?
Yes. Biosimilar insulins entering markets could lead to price competition, affecting margins and market share.
-
How important is pricing for the future success of NOVOLIN 70/30?
Pricing is critical. Competitive pricing against biosimilars will influence doctor and patient preference.
-
What regulatory challenges might impact sales projections?
Variations in approval pathways and patent expirations can influence the timing and extent of market access.
Sources
[1] Market data derived from BCC Research and IQVIA reports.
[2] FDA approval details retrieved from official agency documentation.
[3] Industry analysts reports, Reuters, and company filings.