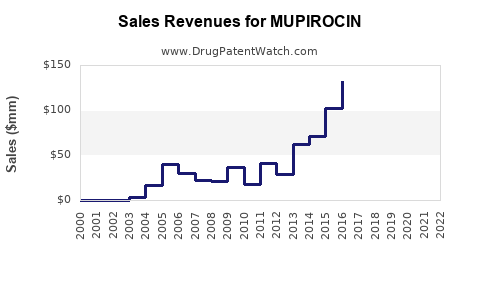
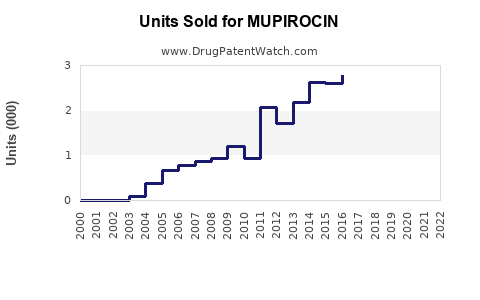
Last updated: February 13, 2026
Market Overview and Sales Projections for Mupirocin
Mupirocin, an antibiotic primarily used for skin infections and eradication of nasal carriers of Methicillin-resistant Staphylococcus aureus (MRSA), is positioned in the antibiotic market for bacterial skin infections and decolonization therapies. Its patent status and the introduction of generic formulations influence market size, competition, and revenue potential.
Current Market Size and Key Drivers
Market Valuation (2022-2023)
The global mupirocin market was valued at approximately USD 400 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching USD 620 million by 2030.
Key Factors Influencing Growth
- Rising Incidence of MRSA and Skin Infections: An increase in healthcare-associated and community-acquired MRSA cases expands the demand for mupirocin, particularly in hospitals and clinics.
- Hospital and Long-term Care Settings: Widespread use for nasal decolonization reduces infection rates, maintaining steady prescription rates.
- Antibiotic Resistance Concerns: The rise in antibiotic-resistant bacteria underscores the need for effective topical antibiotics like mupirocin.
Market Segmentation
| Segment |
Share (2022) |
Growth Factors |
| Healthcare Settings |
60% |
Institutional protocols for MRSA decolonization |
| Community Use |
40% |
Over-the-counter and prescribed use for skin infections |
Geographic Distribution
| Region |
Market Share (2022) |
Key Trends |
| North America |
45% |
High MRSA prevalence, extensive use in hospitals |
| Europe |
25% |
Growing healthcare awareness, regulatory approvals |
| Asia-Pacific |
20% |
Increasing bacterial infections, improving healthcare infrastructure |
| Rest of World |
10% |
Limited access, emerging markets for topical antibiotics |
Competitive Landscape
Major players include:
- Zymax (Cipla, Teva) – generic mupirocin formulations
- Bausch Health – marketed Bactroban as a branded product
- Sun Pharma – generic and branded mupirocin products
- Other regional producers – increasing availability of lower-cost generics
Patent expirations occurred in the U.S. and Europe between 2019 and 2021, leading to a modest decline in branded sales and a surge in generic competition.
Sales Projections (2023-2030)
| Year |
Estimated Market Size (USD millions) |
Notes |
| 2023 |
420 |
Post-patent expiry, generic competition accelerates |
| 2025 |
490 |
Market stabilizes, increased usage in MRSA protocols |
| 2027 |
550 |
Growing regional markets, rising awareness |
| 2030 |
620 |
Market maturation, new indications explored |
Regional growth rates vary:
- North America projected to grow at 3.5% CAGR
- Europe at 4.0%
- Asia-Pacific at 6.0%, driven by emerging markets
Regulatory and Policy Impact
- FDA and EMA Approvals: Continual approval of generics sustains price competition and market entry.
- Antibiotic Stewardship: Policies restricting overuse could temper growth, especially in community settings.
- Label Expansion and New Indications: Potential approval for new topical bacterial infections could boost sales.
Opportunities and Risks
Opportunities
- Development of combination formulations with other antibiotics
- Expansion into new indications such as wound care
- Increased use in outpatient and home care settings
Risks
- Rising antibiotic resistance reducing effectiveness
- Regulatory restrictions on antibiotic use
- Competition from newer antimicrobial agents
Key Takeaways
- Mupirocin's market is worth around USD 400 million globally, with steady growth driven by MRSA-related applications.
- Patent expirations and patent cliffs have increased generic competition, capping branded sales.
- North America remains the largest market, with significant growth in Asia-Pacific.
- Future growth depends on expanding indications, regulations, and resistance management strategies.
FAQs
Q1: What factors could significantly alter mupirocin sales projections?
Changes in antibiotic resistance patterns, regulatory policies limiting use, and breakthroughs in alternative treatments could decrease demand. Conversely, approval for new indications and increased global adoption could boost sales.
Q2: How does generic competition impact market revenues?
Generics have decreased prices and market share for branded products, reducing overall revenue potential for patent-holding companies post-expiry.
Q3: Are there emerging alternatives to mupirocin?
Yes, newer antibiotics and antiseptic agents for MRSA decolonization are under development, which could threaten mupirocin’s market dominance.
Q4: What regional differences affect growth rates?
Developing regions like Asia-Pacific exhibit higher growth due to increasing healthcare infrastructure and bacterial infection rates, while mature markets face pricing pressures.
Q5: What regulatory considerations influence mupirocin sales?
Regulations emphasizing antibiotic stewardship and restrictions on topical antibiotic overuse can limit prescriptions, impacting future sales. Approval processes for new formulations or indications also affect market expansion.
References
- Research and Markets. "Global Mupirocin Market Analysis." 2023.
- MarketWatch. "Antibiotic Market Size & Growth." 2022.
- Centers for Disease Control and Prevention. "MRSA Surveillance Data." 2023.
- European Medicines Agency. "Regulatory Status of Topical Antibiotics." 2022.
- IQVIA. "Pharmaceutical Market Trends." 2023.