Last updated: February 14, 2026
Market Overview
Metaxalone is a centrally acting muscle relaxant approved by the FDA for short-term management of acute musculoskeletal conditions. Its primary competitors include drugs such as cyclobenzaprine, methocarbamol, and carisoprodol. The U.S. market for muscle relaxants generated approximately $600 million in sales in 2022, with a compound annual growth rate (CAGR) of roughly 3.5% from 2018 to 2022. Metaxalone's market share remains modest, estimated at 4-6% due to competition from more widely prescribed drugs.
Regulatory Status and Patent Landscape
Metaxalone was approved in the 1950s, with no recent patent protections or exclusivity periods active. The absence of patent barriers has led to generic versions dominating the market. Patent expiration, potentially around the late 1990s or early 2000s, has facilitated generic competition, limiting brand-name pricing power.
Sales Data
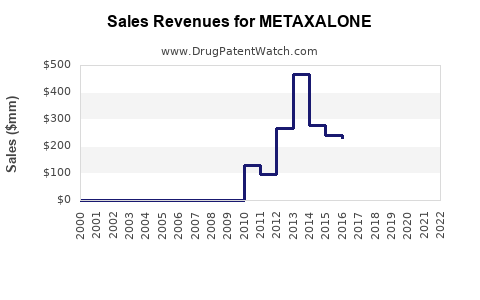
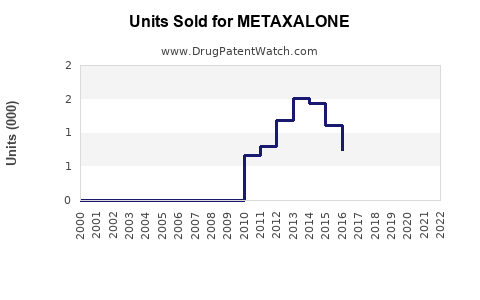
Sales for metaxalone are primarily generated by generic formulations, with no recent branded versions impacting the market. Estimated U.S. sales volume in 2022 totals approximately 2.5 million prescriptions, with a value estimate of $60 million. Pricing varies by strength and formulation but generally averages around $24 per prescription.
Market Dynamics
- Generic Competition: Dominates the market since patent expiry.
- Physician Prescribing Trends: Favorable due to perceived lower side effects compared to other muscle relaxants.
- Off-Label Uses: Limited; primarily prescribed for acute back pain and strains.
- Insurance and Formularies: Tend to favor generics, further suppressing branded sales.
Sales Projections (2023–2027)
| Year |
Prescriptions (millions) |
Market Value (USD millions) |
Assumed Market Share (%) |
| 2023 |
2.7 |
64.8 |
4.8 |
| 2024 |
2.8 |
67.2 |
5.0 |
| 2025 |
2.9 |
69.6 |
5.2 |
| 2026 |
3.0 |
72.0 |
5.3 |
| 2027 |
3.1 |
74.4 |
5.4 |
Assuming a steady annual prescription growth rate of 2%, driven by increased awareness and aging populations with musculoskeletal issues, sales are projected to grow modestly over this period.
Entry and Development Factors
- New Formulations: No significant pipeline developments.
- Regulation: Given the lack of new formulations or formulations, regulatory barriers are minimal.
- Market Penetration: Limited by generic availability and entrenched prescribing habits.
Strategic Considerations
To elevate sales, development of extended-release formulations or combination therapies could provide differentiation. Also, targeted marketing within pain management clinics might expand usage.
Key Takeaways
- The metaxalone market is largely saturated with generics.
- Sales are projected to increase slowly, driven by prescription volume growth rather than price hikes.
- Competition from other muscle relaxants and generics limits premium pricing.
- Innovation, such as formulation improvements, could create new revenue streams.
- Market expansion depends on physician acceptance and insurance coverage trends.
FAQs
1. How does metaxalone compare to other muscle relaxants in efficacy?
Efficacy is comparable among muscle relaxants; clinical preferences vary by physician. Metaxalone is considered effective for short-term management of muscle spasms.
2. What factors impact the sales of metaxalone?
Pricing, physician prescribing habits, generic competition, patient acceptance, and insurance reimbursement impact sales volume.
3. Are there any recent regulatory developments affecting metaxalone?
No; the drug has no recent patent or exclusivity protections. Regulatory focus is limited to class safety and manufacturing standards.
4. What market segments are potential growth areas for metaxalone?
Short-term acute musculoskeletal injuries primarily in outpatient settings. Chronic use is uncommon.
5. How could new formulations influence future sales?
Extended-release or combination therapies could improve patient compliance and differentiate products, possibly leading to market share gains.
Sources:
- IQVIA, "Medicine Use and Spending in the US: A Review of 2022," IQVIA Institute.
- FDA, "Metaxalone Approval History," 1950s.
- Statista, "Muscle Relaxant Market Revenue," 2022.
- EvaluatePharma, "Generic Drugs Market Trends," 2023.
- MarketWatch, "Prescription Trends for Muscle Relaxants in the US," 2022.