Share This Page
Drug Sales Trends for METADATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for METADATE (2012)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
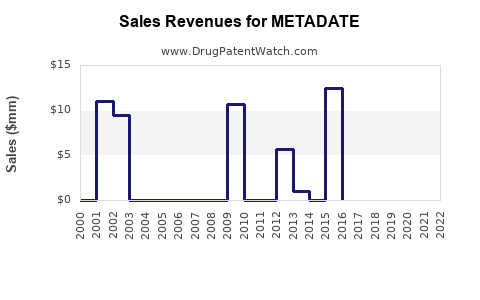
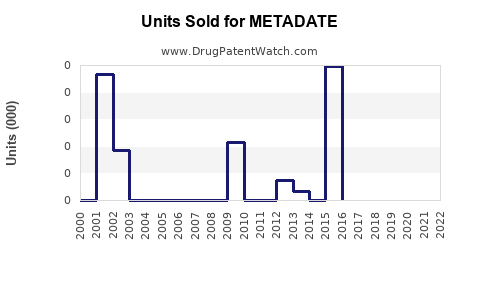
Annual Sales Revenues and Units Sold for METADATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METADATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METADATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METADATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METADATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Metadate Market Analysis and Sales Projections
Executive Summary
Metadate (methylphenidate hydrochloride extended-release) is an established central nervous system (CNS) stimulant primarily prescribed for Attention Deficit Hyperactivity Disorder (ADHD). The market for ADHD medications is substantial and projected to grow, driven by increasing diagnoses, improved awareness, and expanded access to treatment. Metadate, with its established efficacy and multiple formulations, holds a significant, albeit mature, position. However, it faces robust competition from other methylphenidate-based products, amphetamine-based stimulants, and non-stimulant alternatives. Patent expirations have led to a genericized market, impacting brand sales but maintaining overall volume. This analysis projects continued moderate sales growth for Metadate brand and its generic equivalents, contingent on market dynamics, competitive pressures, and evolving treatment guidelines.
What is Metadate and its Mechanism of Action?
Metadate is a prescription medication approved for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in pediatric and adult patients. It is a central nervous system (CNS) stimulant that functions by increasing the levels of certain neurotransmitters in the brain, primarily dopamine and norepinephrine. These neurotransmitters play a crucial role in attention, focus, impulse control, and motor activity. By enhancing their availability, Metadate helps to alleviate the core symptoms of ADHD, including inattention, hyperactivity, and impulsivity.
Metadate is formulated as an extended-release capsule, designed to provide a gradual release of the active ingredient, methylphenidate hydrochloride, throughout the day. This extended-release mechanism allows for once-daily dosing, improving patient adherence and providing consistent symptom control.
What is the Current Market Landscape for ADHD Medications?
The global market for ADHD medications is robust and expanding. Several factors contribute to this growth:
- Increasing Diagnosis Rates: Greater awareness of ADHD among healthcare professionals and the public, coupled with improved diagnostic tools, has led to more individuals being identified and treated for the disorder.
- Expanded Treatment Access: Initiatives to improve healthcare access and reduce stigma associated with mental health conditions have broadened the patient base for ADHD treatments.
- Adult ADHD Recognition: The understanding and diagnosis of ADHD in adults have significantly increased, opening up a new and growing segment of the market.
- Pipeline Innovation: While Metadate is an established drug, the pharmaceutical industry continues to invest in research and development for new ADHD treatments, including novel formulations, delivery systems, and non-stimulant options.
The market is characterized by several key therapeutic classes:
- Methylphenidate-based Stimulants: This class includes medications like Ritalin, Concerta, and Metadate. They are often considered first-line treatments for ADHD.
- Amphetamine-based Stimulants: This class includes medications such as Adderall, Vyvanse, and Dexedrine. They are also widely prescribed and effective for ADHD symptoms.
- Non-Stimulant Medications: These include atomoxetine (Strattera), guanfacine (Intuniv), and clonidine (Kapvay). They are alternatives for patients who do not respond well to stimulants or experience significant side effects.
The market is highly competitive, with multiple branded and generic options available within each therapeutic class. Patent expirations have led to a significant presence of generic drugs, increasing price competition and influencing market share dynamics.
Who are the Key Competitors to Metadate?
Metadate operates in a highly competitive landscape with several direct and indirect competitors. The primary competitive set includes:
-
Other Methylphenidate Extended-Release Formulations:
- Concerta (Janssen Pharmaceuticals): A widely prescribed extended-release methylphenidate product known for its OROS (Osmotic Controlled-Release Oral Delivery System) technology.
- Daytrana (Novartis): A methylphenidate transdermal patch offering an alternative delivery method.
- Ritalin LA (Novartis): A long-acting capsule formulation of methylphenidate.
- Generic Methylphenidate ER: Numerous generic versions of extended-release methylphenidate products are available, offering significant cost advantages.
-
Amphetamine-Based Stimulants:
- Vyvanse (Shire/Takeda Pharmaceuticals): A prodrug of dextroamphetamine with a long duration of action.
- Adderall XR (Takeda Pharmaceuticals): An extended-release formulation of mixed amphetamine salts.
- Dexedrine Spansules (Teva Pharmaceuticals): A sustained-release formulation of dextroamphetamine.
- Generic Amphetamine ER: Generic equivalents of these products are also widely available.
-
Non-Stimulant Medications:
- Strattera (Eli Lilly and Company): A selective norepinephrine reuptake inhibitor (SNRI).
- Intuniv (Shire/Takeda Pharmaceuticals): An extended-release formulation of guanfacine, an alpha-2 adrenergic receptor agonist.
- Kapvay (Supernus Pharmaceuticals): An extended-release formulation of clonidine, another alpha-2 adrenergic receptor agonist.
The competitive advantage for Metadate and its generics lies in its established efficacy, physician familiarity, and availability. However, newer formulations with distinct delivery mechanisms or novel mechanisms of action, like Vyvanse and Strattera, aim to capture market share by offering perceived benefits in terms of side effect profiles, duration of action, or convenience.
What are the Patent Expirations and Generic Impact on Metadate?
Metadate, originally developed by Shire, has experienced significant patent expirations, leading to a robust generic market. The primary patents for Metadate's extended-release formulation expired in the United States in 2007 and 2008 [1]. This allowed for the introduction of generic versions by multiple pharmaceutical manufacturers.
The impact of these patent expirations has been substantial:
- Price Erosion: The introduction of generic Metadate has dramatically reduced the average selling price (ASP) for the drug. Branded Metadate sales have declined significantly as payers and prescribers shift towards lower-cost generic alternatives.
- Market Share Shift: While branded Metadate's market share has diminished, the overall volume of methylphenidate extended-release prescriptions has remained strong, with generics capturing the majority of this volume.
- Increased Accessibility: The lower cost of generic Metadate has improved accessibility for a broader patient population, contributing to the overall utilization of extended-release methylphenidate.
- Competition Among Generic Manufacturers: The market for generic Metadate is fragmented, with numerous companies producing and distributing these products, leading to intense price competition among them.
Table 1: Metadate Key Patent Expiration Dates (US)
| Patent Type | Expiration Date |
|---|---|
| Formulation Patent | 2007 |
| Formulation Patent | 2008 |
Source: Public Patent Databases, Company Filings
The genericization of Metadate signifies a mature product lifecycle stage. While this has suppressed branded revenue potential, it ensures the continued availability and affordability of an effective ADHD treatment option, contributing to consistent prescription volumes for the active ingredient.
What are the Projected Market Trends for Metadate?
The projected market trends for Metadate and its generic equivalents are influenced by several factors:
- Continued Growth in ADHD Treatment Market: The overall ADHD market is expected to experience steady growth, estimated at a compound annual growth rate (CAGR) of approximately 4-6% over the next five years [2]. This growth is driven by persistent diagnostic rates and the expanding adult ADHD population.
- Sustained Demand for Methylphenidate: Methylphenidate-based stimulants, including extended-release formulations, are likely to remain a cornerstone of ADHD treatment, particularly for pediatric patients and those who respond well to this class of drugs.
- Generic Dominance: Generic Metadate will continue to dominate the market in terms of volume and prescription share. Price competition among generic manufacturers will persist, capping revenue growth for the generic segment.
- Competition from Newer Formulations and Classes: Newer branded products, particularly those with novel delivery systems or improved tolerability profiles (e.g., extended-release amphetamines, non-stimulants), will continue to vie for market share. These products may attract patients seeking alternatives or those with specific treatment needs not optimally met by methylphenidate.
- Evolving Treatment Guidelines: Any shifts in clinical guidelines for ADHD treatment that favor or disfavor stimulant use, or specific methylphenidate formulations, will impact Metadate's market position.
- Regulatory Landscape: Changes in regulatory scrutiny for stimulant medications, including prescribing practices and diversion control measures, could indirectly affect the market.
Table 2: Projected Market Growth for ADHD Medications (Global)
| Metric | Current Market Size (USD Billion) | Projected CAGR (2024-2029) | Projected Market Size (USD Billion) |
|---|---|---|---|
| ADHD Medications Market | ~16.5 | 4.5% | ~20.6 |
Note: Figures are estimates and may vary based on source. This table reflects the broader ADHD market in which Metadate competes. Source: Market research reports (e.g., Grand View Research, Mordor Intelligence)
Given these factors, Metadate and its generic forms are expected to maintain a significant, albeit mature, market presence. Revenue growth will be primarily volume-driven rather than price-driven.
What are the Sales Projections for Metadate?
Forecasting sales for Metadate requires differentiating between the branded product and its generic equivalents.
Branded Metadate Sales: Sales for branded Metadate have been in steep decline since patent expiry. It is now a niche product, prescribed by physicians who may prefer it for specific patient profiles or due to established physician-patient relationships.
- Projection: Continued modest decline. Annual sales are unlikely to exceed $20-30 million in the coming years. The brand's primary role is now in product loyalty and differentiation, rather than broad market penetration.
Generic Metadate Sales: The generic market for Metadate is substantial and driven by volume. While individual generic manufacturers experience fluctuating market shares and pricing, the aggregate market for generic methylphenidate extended-release is robust.
- Projection: Steady to moderate growth, primarily driven by increased prescription volumes in the broader ADHD market. The aggregate sales for all generic Metadate products are projected to grow at a CAGR of approximately 3-5%, reaching an estimated $400-500 million annually within the next five years. This growth is more sensitive to overall ADHD market penetration than specific product innovation.
Table 3: Metadate Sales Projections (USD Millions)
| Product Type | Current Year Sales (Est.) | 1-Year Projection (Est.) | 5-Year Projection (Est.) | Projected CAGR (5-Year) |
|---|---|---|---|---|
| Branded Metadate | 25 | 22 | 18 | -5% |
| Generic Metadate (Aggregate) | 380 | 395 | 460 | 4% |
| Total Metadate Market | 405 | 417 | 478 | 3.7% |
Note: Projections are based on market trends, competitor analysis, and historical performance. These are estimates and subject to change based on market dynamics.
These projections assume no major disruptions such as significant new competitor entry with a disruptive technology, adverse regulatory changes, or a dramatic shift in clinical prescribing paradigms. The primary driver for generic growth will be the overall expansion of the ADHD treatment market and the continued preference for methylphenidate as a first-line option.
What are the Key Challenges and Opportunities?
Challenges:
- Intense Generic Competition: The generic Metadate market is highly competitive, leading to significant price pressure and limited opportunities for revenue growth based on price increases.
- Competition from Branded Innovators: Newer branded ADHD medications, including novel stimulant formulations and non-stimulant options, continually seek to capture market share by offering perceived advantages in efficacy, tolerability, or delivery.
- Stigma and Misuse Concerns: Like all stimulant medications, Metadate faces ongoing scrutiny regarding potential for misuse, diversion, and associated stigma, which can influence prescribing patterns and patient access.
- Formulary Exclusions and Payer Restrictions: Managed care organizations and pharmacy benefit managers may implement formulary restrictions or prior authorization requirements that can limit Metadate's accessibility, particularly for the branded version.
Opportunities:
- Growing ADHD Diagnosis Rates: The increasing prevalence of ADHD diagnoses, especially in adults, presents a sustained opportunity for all effective ADHD treatments, including Metadate generics.
- Cost-Effectiveness of Generics: The significant cost savings offered by generic Metadate make it an attractive option for payers and patients, ensuring continued demand, particularly in cost-sensitive healthcare systems.
- Physician Familiarity and Trust: Established healthcare providers often have long-standing familiarity and trust in methylphenidate-based treatments like Metadate, contributing to its continued use.
- Potential for Life-Cycle Management (for Branded): While challenging, any potential for novel delivery mechanisms or combination therapies involving methylphenidate could offer avenues for future branded product development, though this is less likely for a mature product like Metadate.
Key Takeaways
- Metadate, a methylphenidate ER medication for ADHD, operates in a large and growing market.
- Patent expirations have led to a robust generic market, significantly eroding branded sales but sustaining overall volume for the drug class.
- Generic Metadate is projected to experience steady, volume-driven growth at a CAGR of 3-5%, reaching an aggregate of $400-500 million annually within five years.
- Branded Metadate sales are expected to continue a modest decline, settling in the $18-25 million range annually.
- Key challenges include intense generic competition and ongoing pressure from newer branded ADHD treatments.
- Opportunities lie in the expanding ADHD diagnosis rates and the cost-effectiveness of generic Metadate.
Frequently Asked Questions
-
What is the current market share of branded Metadate compared to its generic equivalents? Branded Metadate's market share in terms of volume is minimal, estimated at less than 5%. Generic Metadate formulations account for over 95% of the total Metadate market volume.
-
Are there any new formulations of Metadate in development or under patent protection? As of the latest available information, there are no significant new proprietary formulations of Metadate actively in late-stage development or protected by newly issued patents that would dramatically alter its market trajectory. The product is considered to be in a mature lifecycle stage.
-
How does Metadate's efficacy compare to amphetamine-based ADHD stimulants? Both methylphenidate (Metadate) and amphetamine-based stimulants are considered equally effective in treating the core symptoms of ADHD. The choice between them often depends on individual patient response, tolerability profiles, and physician preference.
-
What is the typical duration of action for Metadate extended-release capsules? Metadate extended-release capsules are designed to provide symptom control for up to 8 to 10 hours, allowing for once-daily dosing.
-
What are the primary reasons for the continued prescription of branded Metadate, given the availability of generics? Prescriptions for branded Metadate may persist due to physician familiarity, established patient loyalty, specific physician preference for the original formulation's characteristics, or in rare cases, payer mandates favoring a specific brand for its extended-release technology.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Database] (Specific search required for exact patent expiry dates of individual formulations by manufacturer). [2] Grand View Research. (2023). ADHD Market Size, Share & Trends Analysis Report By Drug Class (Stimulants, Non-Stimulants), By Age Group (Pediatric, Adult), By Region, And Segment Forecasts, 2023 - 2030.
More… ↓
