Last updated: February 19, 2026
Summary: LUNESTA (eszopiclone), a non-benzodiazepine hypnotic, is nearing the end of its patent exclusivity period, creating significant market opportunities for generic manufacturers. This analysis details LUNESTA's patent history, current market status, and projects the impact of generic entry on its sales and market share. The drug's established efficacy in treating insomnia, coupled with the impending loss of market exclusivity, indicates a substantial shift in the competitive landscape and pricing dynamics.
What is LUNESTA and its Therapeutic Indication?
LUNESTA, developed by Sun Pharmaceutical Industries (previously Sepracor), is an eszopiclone-based medication. It belongs to a class of drugs known as non-benzodiazepine hypnotics, or "Z-drugs." LUNESTA is prescribed for the treatment of insomnia, characterized by difficulties with sleep onset and/or sleep maintenance. The drug functions by enhancing the effect of gamma-aminobutyric acid (GABA), a neurotransmitter that inhibits brain activity, thereby promoting sleep. LUNESTA is available in various dosage strengths, including 1 mg, 2 mg, and 3 mg tablets.
What is LUNESTA's Patent Exclusivity Status?
The primary patent for LUNESTA, U.S. Patent No. 5,929,070, covering the compound eszopiclone, was issued on July 27, 1999. This patent was set to expire on April 16, 2015. Supplementary patents and regulatory exclusivities also contributed to the drug's market protection.
- U.S. Patent No. 5,929,070:
- Issue Date: July 27, 1999
- Expiration Date: April 16, 2015
- Orphan Drug Exclusivity: LUNESTA received seven years of orphan drug exclusivity from the U.S. Food and Drug Administration (FDA) for its indication in treating primary insomnia in patients aged 65 and older due to its potential use in elderly patients who may have difficulty metabolizing the drug. This exclusivity ended on April 20, 2008.
- New Chemical Entity (NCE) Exclusivity: LUNESTA was granted five years of NCE exclusivity, which expired in December 2005.
- Patent Litigation and Generic Entry: The expiration of the primary patent was followed by significant patent litigation. Numerous generic manufacturers filed Abbreviated New Drug Applications (ANDAs) challenging the validity and enforceability of LUNESTA's patents. Legal battles resulted in several key rulings.
- In 2009, a U.S. District Court ruled that LUNESTA's key patent was invalid, paving the way for generic competition. This decision was later upheld on appeal.
- The first generic versions of eszopiclone entered the U.S. market in early 2010.
What is LUNESTA's Market Performance and Sales History?
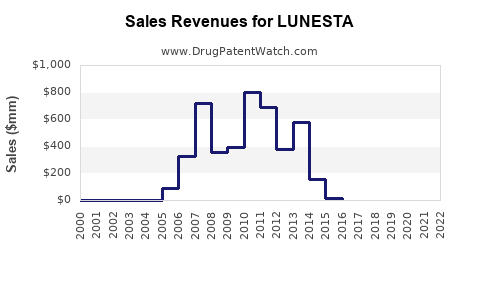
Prior to the widespread availability of generics, LUNESTA achieved significant market success. Its unique chemical structure (the S-enantiomer of zopiclone) and marketing efforts contributed to its strong sales performance.
| Year |
U.S. Net Sales (in millions USD) |
| 2006 |
$711.9 |
| 2007 |
$832.3 |
| 2008 |
$876.3 |
| 2009 |
$820.1 |
| 2010 |
$462.3 |
| 2011 |
$290.1 |
| 2012 |
$195.9 |
| 2013 |
$130.8 |
| 2014 |
$89.2 |
| 2015 |
$65.5 |
Source: Sun Pharmaceutical Industries Annual Reports, SEC Filings.
The sharp decline in sales from 2010 onwards is directly attributable to the introduction of generic eszopiclone. The market for prescription sleep aids is highly competitive, and the availability of lower-cost generic alternatives rapidly erodes the market share of branded drugs.
What is the Competitive Landscape for Sleep Medications?
The sleep aid market is characterized by a broad range of therapeutic options, including benzodiazepines, non-benzodiazepine hypnotics, and over-the-counter (OTC) remedies. LUNESTA competes within the prescription sedative-hypnotic category. Key competitors historically and currently include:
- Zolpidem (Ambien, Ambien CR, generic): A widely prescribed non-benzodiazepine hypnotic.
- Zaleplon (Sonata, generic): Another non-benzodiazepine hypnotic with a shorter half-life.
- Eszopiclone (Lunesta, generic): The active pharmaceutical ingredient of LUNESTA.
- Temazepam (Restoril, generic): A benzodiazepine used for insomnia.
- Other newer agents: While LUNESTA was a prominent player, newer medications and treatment modalities continue to emerge.
The market dynamics are heavily influenced by:
- Pricing: Generic drugs offer a significant cost advantage, making them the preferred choice for many payers and patients.
- Efficacy and Safety Profiles: While LUNESTA demonstrated efficacy, concerns regarding dependence, withdrawal, and side effects are present across the class.
- Prescriber Preferences and Guidelines: Clinical guidelines and physician prescribing habits influence drug selection.
- Payer Formularies: Insurance company formularies often favor drugs with lower cost and established efficacy, including generics.
What are the Projected Market Impacts of LUNESTA's Patent Expiration?
The expiration of LUNESTA's primary patent and subsequent generic entry have fundamentally reshaped its market position.
- Price Erosion: Branded LUNESTA has experienced dramatic price reductions as generic eszopiclone has gained market share. This trend is typical for prescription drugs once patent protection ceases.
- Market Share Shift: The majority of LUNESTA's prescription volume has transitioned to generic eszopiclone. Brand loyalty for LUNESTA is minimal in the face of significantly cheaper generic alternatives.
- Generic Manufacturer Opportunity: The market is now dominated by generic eszopiclone manufacturers, competing primarily on price and distribution channels.
- Continued Demand for Eszopiclone: While the brand LUNESTA's sales have plummeted, the demand for the active compound eszopiclone remains, now fulfilled by generic products. The total market size for eszopiclone (including branded and generic) has been significantly impacted by price compression rather than a decline in patient utilization.
Projected Market Share (Eszopiclone Compound):
| Market Segment |
Projected Share (2024-2026) |
Notes |
| Generic Eszopiclone |
95-98% |
Dominant segment due to cost-effectiveness. |
| Branded LUNESTA |
2-5% |
Niche market, primarily for patients with specific payer coverage or preferences. |
Sales Projections for Branded LUNESTA:
Given the established generic competition and significant market share transfer, branded LUNESTA sales are projected to remain at minimal levels, continuing their downward trend.
| Year |
Projected U.S. Net Sales (Branded LUNESTA, in millions USD) |
| 2024 |
$5-10 |
| 2025 |
$3-7 |
| 2026 |
$2-5 |
These projections assume no significant market disruptions or new indications for the branded product.
What are the Key Takeaways?
- LUNESTA's core patent expired in April 2015, leading to the introduction of generic eszopiclone.
- Generic eszopiclone entered the market in early 2010, initiating a rapid decline in branded LUNESTA sales.
- The market for eszopiclone is now overwhelmingly dominated by generic manufacturers, with price competition being the primary driver.
- Branded LUNESTA sales have significantly diminished and are projected to remain at very low levels.
- The demand for eszopiclone as a treatment for insomnia persists, now met by cost-effective generic alternatives.
Frequently Asked Questions
-
When did the primary patent for LUNESTA expire in the United States?
The primary U.S. patent for LUNESTA (U.S. Patent No. 5,929,070) expired on April 16, 2015.
-
When did generic versions of eszopiclone first become available in the U.S.?
The first generic versions of eszopiclone entered the U.S. market in early 2010.
-
What is the current market share of branded LUNESTA compared to generic eszopiclone?
Generic eszopiclone holds an estimated 95-98% of the market, with branded LUNESTA accounting for a minimal 2-5% share.
-
What factors contributed to the rapid decline in LUNESTA's sales after patent expiration?
The introduction of lower-cost generic eszopiclone, combined with the competitive nature of the sleep aid market and payer preferences for cost-effective options, led to the swift decline in branded LUNESTA sales.
-
Are there any other significant patents or exclusivities that protected LUNESTA?
Yes, LUNESTA had a five-year New Chemical Entity (NCE) exclusivity that expired in December 2005, and a seven-year orphan drug exclusivity for elderly patients, which expired in April 2008. However, the compound patent expiration was the most significant factor enabling broad generic entry.
Citations
[1] Sun Pharmaceutical Industries. (Various Years). Annual Reports and SEC Filings. U.S. Securities and Exchange Commission.
[2] U.S. Patent and Trademark Office. (1999). U.S. Patent No. 5,929,070. Washington, D.C.
[3] Court documents related to patent litigation for eszopiclone. (Various years). U.S. District Courts and Circuit Courts.