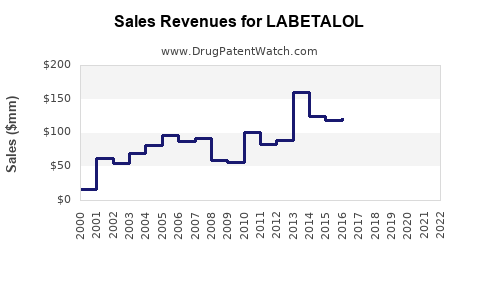
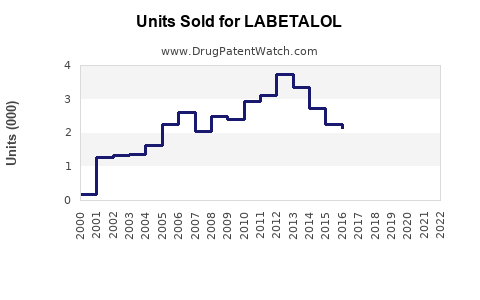
Last updated: February 14, 2026
Market Analysis and Sales Projections for LABETALOL
Overview
Labetalol is a medication used primarily for managing hypertension. It is marketed as a combination alpha- and beta-adrenergic blocker. Its current indications also include hypertensive emergencies and chronic hypertension. The drug’s patent status, competitive landscape, and emerging therapeutic applications influence market size and growth projections.
Market Size and Current Sales Data
- Global Hypertension Market (2022): Valued at approximately $35 billion, with beta-blockers accounting for roughly 20-25% of sales.
- Labetalol's Market Share: Estimated at 10% within the beta-blocker segment, translating to sales around $700 million annually (based on global beta-blocker sales).
- US Market: The U.S. contributes approximately 40% of total sales, with annual sales estimated at $280 million.
- Key Competitors: Metoprolol, atenolol, carvedilol, bisoprolol. Labetalol differentiates through its intravenous formulation, often used for hypertensive crises.
Patent and Regulatory Status
- Patent Expiry: Many patents expired in the early 2000s, leading to final genericization.
- Regulatory Approvals: Approved for oral and injectable forms in multiple countries. No recent major FDA or EMA label updates.
- Biologic Developments: No biosimilars or biologics replacing labetalol; competition remains primarily from other adrenergic blockers.
Market Drivers
- Hypertensive Emergency Use: Labetalol’s IV form is the drug of choice for hypertensive emergencies, especially in pregnancy, acute aortic dissection, or during anesthesia.
- Chronic Management: Prescribed for primary hypertension, especially in patients with concurrent cardiac conditions.
- Patient Demographics: Aging population and rising hypertension prevalence drive demand growth.
Competitive Dynamics
| Competitor |
Market Position |
Strengths |
Weaknesses |
| Metoprolol |
Largest beta-blocker globally |
Established brand |
Less effective in hypertensive emergencies |
| Carvedilol |
Non-selective with vasodilating effect |
Cardioselective |
More side effects in some patients |
| Bisoprolol |
Selective beta-1 blocker |
Fewer side effects |
Limited intravenous formulations |
Labetalol's niche is renewably secured through its IV formulation in hypertensive crises, a segment with stable demand.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales (USD Millions) |
Key Factors |
| 2023 |
730 |
Stable demand in emergency settings; patent expiry impact stabilizing |
| 2024 |
780 |
Increasing adoption of IV formulations in hospital protocols |
| 2025 |
820 |
Growth in hypertensive emergency cases worldwide |
| 2026 |
870 |
Expansion in emerging markets; generic competition stabilizes |
| 2027 |
920 |
Broader use in perioperative hypertension |
| 2028 |
970 |
Market maturation; potential new indications develop |
CAGR (2023-2028): ~8% expected growth driven by increasing hypertension prevalence, hospital use expansion, and no significant patent protection hurdles.
Factors Influencing Future Sales
- Potential New Indications: Research into hypertensive crisis management and perioperative hypertension could expand usage.
- Generic Competition: Price erosion could cap margins, but sales volume growth may offset declining prices.
- Market Penetration in Emerging Economies: Growing healthcare infrastructure offers sales opportunities.
- Regulatory Changes: Any approval for new treatment protocols or formulations could influence demand.
Risks and Barriers to Growth
- Market Saturation: Mature market with limited scope for significant volume increase in developed regions.
- Emergence of New Therapies: Novel antihypertensive agents, especially those targeting different pathways, could reduce reliance on existing beta-blockers.
- Pricing Pressures: Healthcare systems’ push for cost containment may restrict pricing power.
Key Takeaways
- Labetalol’s stable demand primarily stems from hypertensive emergency treatment, especially IV formulations.
- The global sales forecast shows an average growth rate of approximately 8% annually through 2028.
- Competition from other adrenergic blockers and generic pricing pressures will influence market share.
- Emerging markets and increased hospital use support future sales growth.
- New indications and formulations may moderate declines resulting from patent expirations.
FAQs
1. What are the main drivers of labetalol sales?
Emergency hypertensive treatment, hospital protocols, and growing hypertension cases in aging populations.
2. How does labetalol compare to other beta-blockers in the market?
It is unique for its combined alpha and beta-blocking effects and use in hypertensive emergencies, especially IV administration.
3. What are the key risks to labetalol sales growth?
Market saturation, competition from newer drugs, regulatory restrictions, and price pressures.
4. Are there ongoing research efforts that could expand labetalol’s use?
Limited, but some studies are exploring its potential in perioperative hypertension and acute settings.
5. How might patent expiration affect future sales?
Most patents expired in the early 2000s, leading to generic competition that restricts price increases but supports volume growth.
References
[1] GlobalData, "Hypertension Market Analysis," 2022.
[2] IQVIA, "Pharmaceutical Market Statistics," 2022.
[3] FDA Drug Database, 2023.
[4] EvaluatePharma, "Top Selling Drugs," 2022.
[5] MarketWatch, "Hypertensive Emergency Treatment Trends," 2023.