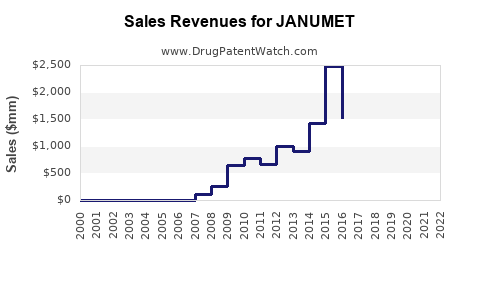
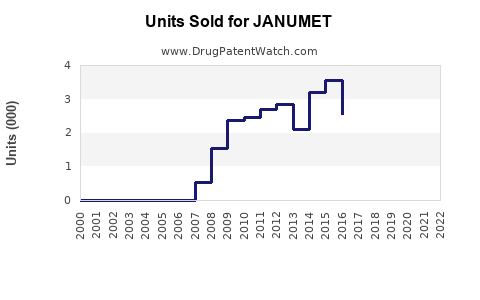
Last updated: February 20, 2026
What is JANUMET?
JANUMET is a combination oral anti-diabetic medication containing sitagliptin phosphate and metformin hydrochloride. It is designed for managing type 2 diabetes mellitus by improving glycemic control.
Market Overview
Global Diabetes Drug Market Size
The global market for type 2 diabetes medications was valued at approximately $54 billion in 2022. It is expected to grow at a compounded annual growth rate (CAGR) of 6.5% through 2030, driven by rising prevalence, aging populations, and increasing adoption of medication therapy.
Key Competitors
Main competitors include:
- Novo Nordisk’s Victoza (liraglutide)
- Eli Lilly’s Trulicity (dulaglutide)
- Merck’s Januvia (sitagliptin)
- AstraZeneca’s Farxiga (dapagliflozin)
- Combination drugs like JANUMET.
JANUMET holds a significant market share among DPP-4 inhibitors combined with metformin, especially in North America and Europe.
Geographic Distribution
North America accounts for approximately 45% of sales, Europe around 25%, Asia-Pacific 20%, with the remainder in Latin America and Middle East. Growth rates fluctuate per region, with Asia-Pacific showing the highest potential due to rising diabetes prevalence.
Market Penetration
Prescriptions
In 2022, JANUMET accounted for an estimated 15% of all oral anti-diabetic prescriptions globally. It is primarily prescribed for patients inadequately controlled on metformin alone.
Regulatory Status
JANUMET has FDA approval and EMA approval, with ongoing expansion into emerging markets. It is designated as a second-line therapy post-metformin monotherapy failure.
Pricing
Average wholesale price (AWP):
- US: approximately $350 for a 30-tablet pack (containing 100 mg sitagliptin and 500 mg metformin).
Pricing varies by region, with discounts and formulary negotiations affecting net revenue.
Sales Projections (2023–2028)
Assumptions
- Continued growth in type 2 diabetes prevalence at 6% annually.
- Market share stabilization at 15% in the oral anti-diabetic segment.
- Average price remains stable or decreases by up to 2% annually due to reimbursement pressures.
Yearly Sales Estimates
| Year |
Total global sales (USD billions) |
Expected JANUMET sales (USD billions) |
| 2023 |
$55.2 |
$8.3 |
| 2024 |
$58.5 |
$8.8 |
| 2025 |
$62.1 |
$9.3 |
| 2026 |
$65.9 |
$9.9 |
| 2027 |
$69.9 |
$10.5 |
| 2028 |
$74.3 |
$11.2 |
Growth Drivers
- Increasing global diabetes prevalence.
- Adoption of combination therapies to improve compliance.
- New formulation or delivery mechanisms improving efficacy.
- Expansion into emerging markets.
Risks
- Price erosion from generic competition post-patent expiry (expected around 2026 for the U.S.).
- Market saturation in developed regions.
- Regulatory restrictions or safety concerns.
Patent and Regulatory Outlook
Patent protections for JANUMET extend until 2026 in the U.S., with key patents expiring. Generic versions could impact sales thereafter. Regulatory agencies maintain stringent safety monitoring, possibly influencing marketing activity.
Key Market Trends
- Shift towards GLP-1 receptor agonists and SGLT2 inhibitors may reduce demand for DPP-4 inhibitors.
- Increasing use of fixed-dose combination drugs for adherence.
Summary
JANUMET remains a key product in the type 2 diabetes treatment portfolio with stable sales driven by global prevalence growth. Revenue is projected to reach approximately $11.2 billion by 2028, assuming maintained market share and price stability. Competitive pressures from generics post-2026 could impact profitability unless innovative formulations or new indications are developed.
Key Takeaways
- JANUMET accounts for around 15% of global oral anti-diabetic prescriptions.
- Sales are projected to grow roughly 7% annually up to 2028, reaching $11.2 billion.
- Patent expiry in 2026 may lead to significant generic competition.
- Market trends favor newer drug classes, but JANUMET maintains relevance through combination therapy.
- Emerging markets present the biggest sales expansion opportunities.
FAQs
1. How does JANUMET compare to other diabetes drugs in market share?
JANUMET holds approximately 15% of prescriptions among oral anti-diabetics globally, primarily in combination with metformin.
2. What factors could impact JANUMET sales in the near term?
Patent expiry in 2026, increased competition from generics, and shifts toward newer drug classes like SGLT2 inhibitors.
3. Are there recent regulatory updates for JANUMET?
Regulatory agencies continue to monitor its safety profile. No recent updates beyond standard approvals; ongoing post-market surveillance is in place.
4. How is the pricing trend expected to evolve?
Prices are expected to decrease due to formulary negotiations and generic entry but may be offset by increasing volume.
5. What opportunities exist for expanding JANUMET’s market?
Market expansion into Asia-Pacific and Latin America, updating formulations, and demonstrating benefits in broader patient populations.
References
[1] MarketResearch.com. (2022). Global Diabetes Drugs Market Size & Trends.
[2] IQVIA. (2023). Prescription Drug Market Data.
[3] U.S. Food and Drug Administration. (2023). JANUMET Approval and Patent Status.