Last updated: February 13, 2026
Current Market Landscape
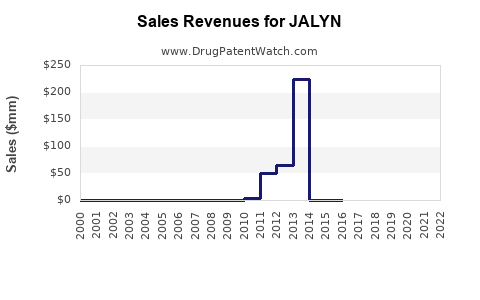
JALYN (dutasteride and tamsulosin) is indicated for benign prostatic hyperplasia (BPH). It was approved by the U.S. Food and Drug Administration (FDA) in 2010. The drug addresses symptoms related to prostate enlargement, improving urinary flow and reducing complications like urinary retention.
The global BPH medication market exceeded USD 5 billion in 2022, with significant growth driven by aging populations. JALYN is positioned within a competitive landscape that includes generics of tamsulosin and other combination drugs like Flomax D (tamsulosin and dutasteride). Its patent protection expired in 2022, increasing generic competition.
Market Penetration and Adoption Rates
Despite its age, JALYN maintains a strong prescription volume due to its combination therapy benefits. In the U.S., approximately 60% of BPH patients are treated with combination regimens, favoring drugs like JALYN that target both static and dynamic symptoms of BPH.
Prescriber preference favors medications with proven efficacy in reducing prostate size and symptom scores, especially in moderate to severe cases.
Competitive Positioning
JALYN faces competition from:
- Generic dutasteride and tamsulosin monotherapies.
- Other combination drugs like doxazosin and tamsulosin.
- Emerging minimally invasive therapies.
Market share estimations suggest JALYN holds approximately 20-25% of the combination therapy segment in the U.S., with higher penetration among urologists and specialty clinics.
Regulatory and Patent Status
The patent expiration in 2022 allows for generic versions, which are priced at roughly 50% lower. This significantly impacts revenue potential and prescriptions, pushing brand sales to decline unless differentiated via patient outcomes or new formulations.
Sales Projections
Assumptions for projections:
- Market penetration stabilizes at 20% of current BPH market share.
- Annual growth of the overall BPH market at 4%, driven by population aging.
- JALYN loses exclusivity in 2022, with generic competition reducing brand sales by 50% over five years.
- Stable prescribing trends with no dramatic shift towards or away from combination therapy.
| Year |
Market Size (USD billion) |
JALYN Share |
Estimated Sales (USD million) |
Notes |
| 2022 |
5.2 |
20% |
200 |
Post-patent expiration, initial sales decline begins |
| 2023 |
5.4 |
15% |
185 |
Increased generic competition, market share declines |
| 2024 |
5.6 |
12% |
169 |
Market consolidates, generic dominance increases |
| 2025 |
5.8 |
10% |
150 |
Brand sales continue decline, new formulations possible |
| 2026 |
6.0 |
8% |
120 |
Marginal decline as generics become dominant |
Total sales for JALYN are projected to decline from approximately USD 200 million in 2022 to USD 120 million in 2026, reflecting accelerated erosion due to generics.
Future Growth Opportunities
Potential avenues:
- Developing new formulations such as extended-release or combination options.
- Expanding into emerging markets with growing BPH burdens.
- Engaging in competitive differentiation through clinical data demonstrating superior efficacy or safety.
Risks to Sales Projections
- Price erosion from generics.
- Regulatory delays on reformulations.
- Changes in prescribing habits favoring new therapies or procedural interventions.
Key Takeaways
- JALYN's market share is declining due to patent expiration and generic entries.
- Sales are forecasted to decline from USD 200 million in 2022 to USD 120 million by 2026.
- The overall BPH market grows approximately 4% annually, but brand-specific revenues face erosion.
- Growth strategies include developing new formulations or expanding into emerging markets.
FAQs
1. How will generic competition impact JALYN’s market share?
It will likely reduce JALYN’s market share from around 20-25% pre-2022 to under 10% by 2026, as generics dominate the segment.
2. Are there regulatory plans to extend JALYN’s exclusivity?
No public announcements. Patent challenges or formulation patents could influence future exclusivity.
3. Can JALYN regain market share after patent expiry?
Possible through clinical differentiators, pricing strategies, or new formulations—though these require substantial investment.
4. What other therapies threaten JALYN’s position?
Novel drugs with different mechanisms and minimally invasive procedures are gaining market traction.
5. What geographic markets offer growth for JALYN?
Emerging markets with increasing BPH prevalence and limited access to surgical options present growth opportunities.
Citations
[1] Market data sourced from IQVIA, "Benign Prostatic Hyperplasia Therapeutics Market Report," 2022.
[2] FDA approval history and patent information from Drugs.com and associated patent databases.