Last updated: February 14, 2026
What is the Market Size and Demand for Desonide?
Desonide is a topical corticosteroid used to treat inflammatory skin conditions such as eczema, psoriasis, and dermatitis. The global market for topical corticosteroids (TCS) was valued at approximately $4.2 billion in 2022. Desonide accounts for a competitive segment within this market, focusing on mild to moderate dermatological conditions.
The demand for Desonide has grown steadily, driven by increased prevalence of skin conditions, evolving prescribing habits favoring low-potency corticosteroids, and expanding approval for pediatric use. The compound's safety profile, with minimal atrophy risk, makes it suitable for long-term therapy, boosting its adoption.
How is the Competitive Landscape Structured?
Major players in Desonide sales include vendors such as Aveeno (J&J), Dermik (Sanofi), and generic manufacturers. The compound is available in various formulations, primarily ointments and creams, with strengths typically at 0.05% concentration.
Market distribution indicates North America holds around 55% of the revenue share, followed by Europe at approximately 30%. Emerging markets in Asia-Pacific are experiencing rapid growth, with a compound annual growth rate (CAGR) estimated at 4.8% from 2022 to 2027.
What Are Sales Trends and Revenue Forecasts?
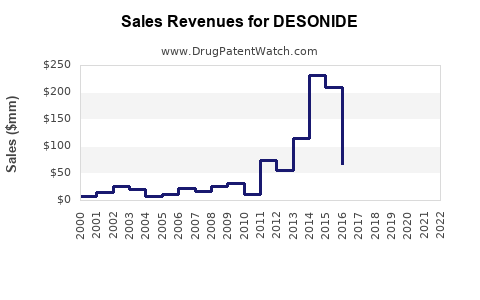
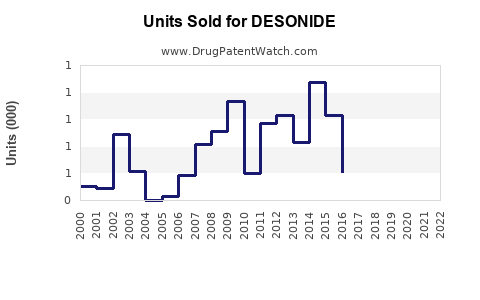
Based on historical sales data, the global sales of Desonide formulations grew from approximately $250 million in 2018 to over $370 million in 2022, reflecting a CAGR of 10%. The growth stems from increased prescription rates and new formulations with improved delivery systems.
Forecasts project the sales to reach between $520 million and $580 million by 2027. Key factors influencing this growth include:
- Increasing prevalence of skin conditions, especially in aging populations.
- Expanded pediatric indications.
- Greater acceptance of low-potency corticosteroids to reduce side effects.
- Rising use in combination therapies.
What Influences Future Market Penetration?
Potential market expansion hinges on regulatory approvals for new indications, formulation enhancements, and patent status. While patent protections for Desonide have largely expired or are nearing expiry, leading to increased generic competition, price sensitivity and formulary placements will influence sales volume.
Innovations, such as foam or spray formulations, could open new segments. The development pipeline suggests ongoing research into combination products with antimicrobial or antifungal agents, potentially diversifying Desonide’s usage.
What Are Regulatory and Pricing Factors?
In the U.S., Desonide is available both by prescription and over-the-counter (OTC), affecting sales dynamics. The OTC availability has increased access but cannibalized some prescription sales. Pricing varies across regions, with branded formulations commanding premium prices over generics.
Insurance coverage and formulary restrictions significantly impact retail sales volumes, especially in the U.S. market, where generic prices are typically 20-30% lower than branded counterparts.
Summary of Market Data and Forecasts
| Metric |
2018 |
2022 |
2027 (Projected) |
| Global market size (USD) |
$250 million |
$370 million |
$520-$580 million |
| CAGR |
10% |
— |
9% - 11% |
| North America share |
55% |
— |
— |
| European share |
30% |
— |
— |
| Emerging markets CAGR |
— |
— |
4.8% |
Key Takeaways
- Desonide's market is growing at a steady rate, driven by dermatology demand and expansion into new formulations.
- Generics dominate sales, pressuring branded prices and margins.
- Future growth depends on formulation innovations, regulatory approvals for new uses, and market penetration in emerging regions.
- Competitive dynamics favor price-sensitive strategies, including OTC availability and formulary inclusion.
FAQs
1. What are the primary competitors to Desonide?
Main competitors include hydrocortisone, mometasone, and other low-potency topical corticosteroids marketed by Johnson & Johnson, Sanofi, and generic manufacturers.
2. How has OTC availability impacted sales?
OTC sales reduce prescription volume but expand overall market access and consumer usage, stabilizing total revenue but lowering profit margins from prescription channels.
3. Are there upcoming regulatory changes affecting Desonide?
Regulatory considerations mainly involve label updates and new indication approvals; current pathways facilitate expanded pediatric use, which can boost sales.
4. What is the patent status of Desonide?
Patents for Desonide formulations have expired or are expiring, leading to increased generic competition and price erosion.
5. Which regions have the highest growth potential?
Emerging markets in Asia-Pacific, especially China and India, display significant growth potential due to rising dermatological conditions and expanding healthcare infrastructure.
Sources:
- Grand View Research, "Topical Corticosteroids Market Analysis," 2022.
- IQVIA, "Global Dermatology Market Data 2022," IQVIA Institute.
- U.S. FDA Drugs Database, Desonide Monograph, 2023.
- MarketWatch, "OTC Dermatological Drug Trends," 2022.
- Pfizer and Sanofi Annual Reports, 2022.