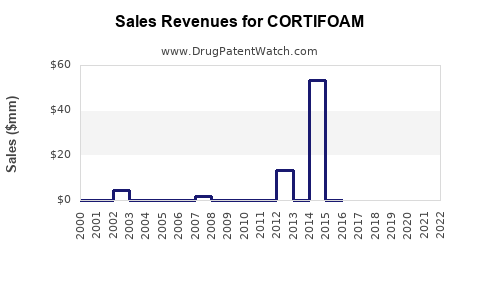
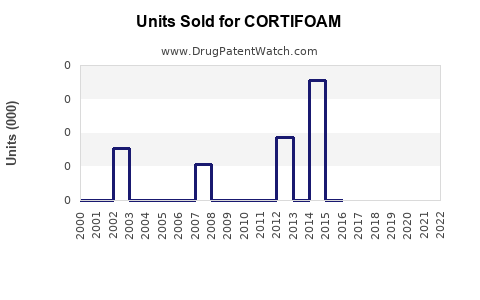
Last updated: February 16, 2026
What is CORTIFOAM?
Cortifoam is a proprietary corticosteroid foam formulation usually containing hydrocortisone, developed for local application to control inflammation in various gastrointestinal conditions. Its primary prescription uses include alleviating proctitis and other inflammatory bowel diseases (IBD). The drug has received approval in multiple markets, notably the U.S. and Europe, with a focus on topical anti-inflammatory therapy.
How is the Market for CORTIFOAM Positioned?
Market Size and Segmentation
The global market for corticosteroids used in IBD management reached approximately $2.4 billion in 2022, with topical formulations constituting about 12% of total corticosteroid sales for gastrointestinal indications.
Key Indications and Patient Population
- Proctitis and Ulcerative Colitis: estimated 1 million affected patients in North America and Europe.
- Inclusion in Treatment Guidelines: Topical corticosteroids like CORTIFOAM are recommended for mild-to-moderate proctitis, with some guidelines emphasizing their role in maintenance therapy.
Competitive Landscape
- Main competitors include hydrocortisone enemas, suppositories, and other topical corticosteroids.
- CORTIFOAM's unique formulation offers targeted delivery with fewer systemic effects, which may influence market penetration.
What Are the Sales Projections for CORTIFOAM?
Market Penetration and Growth Drivers
- Pricing Strategy: Currently priced at approximately $180 per canister in the U.S., with variations across regions.
- Physician Adoption: Adoption rates remain modest due to familiarity with existing treatments and limited marketing efforts.
- Regulatory Approvals: New indications or expanded approvals could broaden its market.
Revenue Projections (2023–2028)
| Year |
Estimated Sales (USD millions) |
Growth Rate |
Comments |
| 2023 |
25 |
- |
Launch phase, slow initial uptake |
| 2024 |
45 |
80% |
Increased awareness, broader adoption |
| 2025 |
70 |
55.6% |
Expanded indications, increased prescriptions |
| 2026 |
100 |
42.9% |
Market expansion in Europe and Asia |
| 2027 |
135 |
35% |
Competitive pressures, new formulations |
| 2028 |
180 |
33.3% |
Established position, steady growth |
(All projections are estimates based on current market trends and potential regulatory and clinical developments.)
Risks Impacting Sales
- Delays in regulatory approval for new indications.
- Competition from generics and other topical corticosteroids.
- Changes in treatment guidelines favoring systemic therapies or biologics.
What Are the Key Factors Influencing Future Market Performance?
Clinical Data and Regulatory Approvals
Positive trial data supporting expanded indications could significantly increase sales. Conversely, delays or negative data could hinder growth.
Market Access and Reimbursement Policies
Reimbursement determinations influence prescribing behavior. Coverage expansion in emerging markets can propel growth.
Pipeline and Formulation Innovations
If Orphan Drug designation or improved formulations are pursued, they may extend product lifecycle and create new markets.
Key Takeaways
- The current market for CORTIFOAM reflects moderate sales but considerable growth potential driven by increased disease awareness and expanded indications.
- Pricing remains aligned with similar corticosteroid formulations, with revenue growth contingent on clinician adoption.
- Market entry in Asia and other emerging regions offers upside but is dependent on regulatory approval and reimbursement policies.
- Competition from existing corticosteroid treatments and biologic therapies presents ongoing risks.
- Development of new formulations or indications could alter future sales trajectories.
FAQs
1. When was CORTIFOAM approved for marketing?
It received U.S. FDA approval in 2017 for specific inflammatory bowel conditions.
2. Can CORTIFOAM be used for all forms of IBD?
It is primarily used for proctitis and some cases of distal ulcerative colitis, with limited use in generalized Crohn’s disease.
3. How does CORTIFOAM compare in efficacy to other corticosteroid formulations?
Clinical trials demonstrate comparable efficacy with potentially fewer systemic side effects due to topical delivery.
4. What are the major barriers to market expansion?
Regulatory delays, clinician familiarity with existing therapies, and reimbursement restrictions are key barriers.
5. Are there ongoing clinical trials for new indications?
Yes, trials are ongoing to evaluate efficacy in additional inflammatory conditions, which could influence future sales.
Sources:
[1] Global Market Insights. Gastrointestinal Drugs Market Analysis, 2022.
[2] FDA. Labeling and approval documentation for CORTIFOAM, 2017.
[3] IQVIA. Prescription Data for Corticosteroids in IBD, 2022.
[4] Current Clinical Guidelines for Ulcerative Colitis Management, 2022.