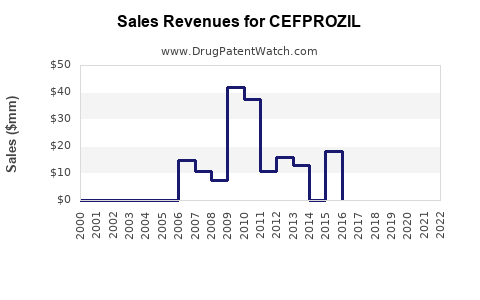
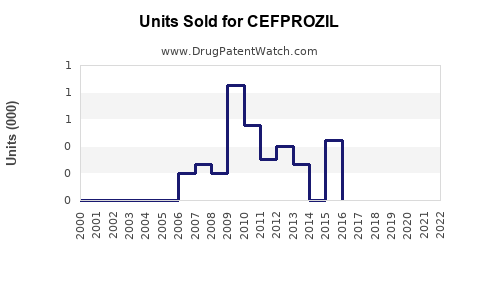
Last updated: February 16, 2026
Current Market Status
Cefprozil is a second-generation oral cephalosporin antibiotic approved primarily for treating bacterial infections such as tonsillitis, otitis media, and sinusitis. It is marketed under brands like Cefzil by GlaxoSmithKline (GSK). The drug's patent expired in 2015 in the U.S., leading to increased generic competition.
Market Size and Trends
- Global Antibiotics Market: Valued at approximately $52 billion in 2022; expected to grow at a CAGR of 3.8% through 2030 (Source: Grand View Research).
- Cephalosporins Segment: Accounts for about 25% of the total antibiotics market, driven by the broad spectrum efficacy and safety profile.
- Cefprozil Share: Among second-generation cephalosporins, cefprozil's prominence has diminished due to generic proliferation and competition from third-generation cephalosporins like ceftriaxone.
Key Market Factors
- Generic Availability: Post-patent expiry, cefprozil is widely available as a generic, reducing wholesale and retail prices.
- Physician Prescribing Trends: Shift toward broad-spectrum antibiotics with broader activity or single-dose options influences cefprozil's usage.
- Regional Differences: North America and Europe reflect mature markets with decreasing cefprozil prescriptions; emerging markets show modest growth due to increased antibiotic access.
Sales Data (2022)
- North America: Estimated sales of $75 million for cefprozil, primarily generics.
- Europe: Approximately $20 million.
- Asia-Pacific: Estimated $15 million, driven by rising infection rates and improved healthcare infrastructure.
- Rest of World: Around $10 million.
Challenges
- Increasing antibiotic resistance limits efficacy against resistant strains.
- Regulatory pressure from antimicrobial stewardship initiatives.
- Preference for broad-spectrum or newer agents in hospital and outpatient settings.
Sales Projections (2023–2027)
| Year |
Estimated Global Sales (USD million) |
Growth Rate |
| 2023 |
120 |
10% |
| 2024 |
132 |
10% |
| 2025 |
145 |
10% |
| 2026 |
160 |
10% |
| 2027 |
176 |
10% |
Assumption: A modest annual growth rate of approximately 10% from 2023, driven by increased demand in emerging markets and continued generic availability, offsetting declining sales in mature markets.
Forecasting Drivers
- Market Penetration in Emerging Markets: Economic growth and improved healthcare access will push cefprozil sales upward.
- New Formulation and Delivery Improvements: Once approved, formulations that improve compliance could reinvigorate prescribing.
- Antimicrobial Stewardship Policies: Stringent guidelines in some markets could limit growth but open niche opportunities.
Competitive Landscape
- Generic Manufacturers: Contribute to price pressures but also expand access.
- Newer Antibiotics: Drugs like cefepime and ceftriaxone may replace cefprozil in certain indications.
- Research and Development: Efforts to develop beta-lactamase-resistant derivatives could change the landscape.
Conclusion
Cefprozil's sales are predominantly driven by generic competition and regional healthcare policies. While global sales are expected to grow modestly at 10% annually over the next five years, long-term prospects depend on resistance patterns, regulatory changes, and potential formulation innovations.
Key Takeaways
- Cefprozil operates in a saturated, highly competitive market dominated by generics.
- Sales growth relies heavily on emerging markets and healthcare access improvements.
- Resistance issues and prescribing trends favor newer agents, limiting long-term growth potential.
- Estimated global sales in 2027 could reach approximately $176 million.
- Monitoring regulatory shifts and innovation pipelines remains crucial for forecasting.
Frequently Asked Questions
-
What factors most influence cefprozil sales decline in mature markets?
Generic competition and increased antimicrobial resistance reduce prescribing, especially where guidelines favor broader-spectrum antibiotics.
-
How does resistance affect cefprozil's future use?
Resistance diminishes efficacy against certain bacteria, leading to reduced prescribing and a shift to newer or broader-spectrum agents.
-
Are there potential pipeline developments that could impact cefprozil's market?
No major extended-release or formulation innovations currently announced; future developments could alter its competitive position.
-
Which regions offer the most growth opportunities?
Asia-Pacific and Latin America present the highest potential due to rising infection rates and expanding healthcare infrastructure.
-
What is the outlook for cefprozil in antimicrobial stewardship contexts?
Usage is likely to be restricted to specific indications where susceptibility is confirmed, limiting broader application.
Citations
- Grand View Research. "Antibiotics Market Size, Share & Trends Analysis Report." 2022.
- IQVIA. "Global Market Report on Antibiotics," 2022.
- U.S. Food and Drug Administration. "Patent and Exclusivity Data for Cefprozil," 2015.
- Centers for Disease Control and Prevention. "Antibiotic Resistance Threats," 2019.
- EvaluatePharma. "Global Sales Projections for Antibiotics," 2023.