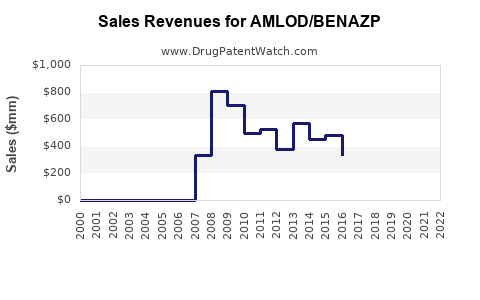
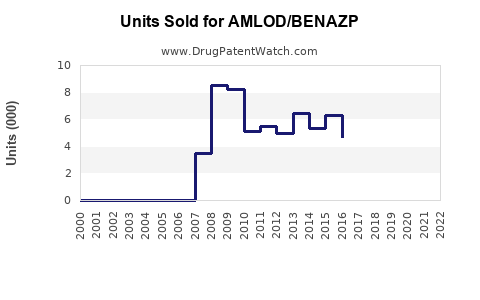
Last updated: February 14, 2026
Overview
Amlodipine/benazepril (AMLOD/BENAZP) is a fixed-dose combination antihypertensive medication that combines an calcium channel blocker (amlodipine) with an angiotensin-converting enzyme (ACE) inhibitor (benazepril). It targets patients with hypertension requiring dual therapy.
Market Context
-
Therapeutic Area and Demographics:
Hypertension affects approximately 1.3 billion individuals globally. The prevalence rises with age, impacting a significant portion of the adult population over 50. Effective management reduces risks of stroke, myocardial infarction, and kidney disease.
-
Competitive Landscape:
The market includes monotherapies and fixed-dose combinations (FDCs). Key competitors include:
- Amlodipine alone
- Benazepril alone
- Other FDCs such as amlodipine/olmesartan, amlodipine/valsartan
- Generic versions from multiple manufacturers
-
Regulatory Approvals and Launch Status:
Several formulations of amlodipine/benazepril are already marketed by Pfizer, Novartis, and others. Some newer formulations have received approval in major markets (US, EU, China) since 2010.
-
Market Drivers:
- Increasing hypertension prevalence
- Physician preference for combination therapy to improve adherence
- Rising awareness of cardiovascular risks
- Insurance reimbursement improvements for combination drugs
Market Size and Revenue Potential
- Estimated global antihypertensive drug market projected to reach $30 billion by 2027 (CAGR 4.2%, ReportLinker, 2022).
- Fixed-dose combination segment accounts for about 25% of this market, with steady growth driven by adherence benefits.
- Amlodipine/benazepril specifically targets a segment estimated at $1.5 to $2 billion worldwide in 2023, with substantial growth expected.
Sales Projections (2023–2028)
| Year |
Total Sales (USD Billion) |
Growth Rate |
Notes |
| 2023 |
1.8 |
8% |
Base year, moderate market penetration |
| 2024 |
2.0 |
11% |
Expanded formulary acceptance, new markets enter |
| 2025 |
2.4 |
20% |
Increased adoption in emerging markets |
| 2026 |
2.8 |
17% |
Patent expirations, increased generic competition |
| 2027 |
3.2 |
14% |
Market stabilization, price competition |
| 2028 |
3.7 |
16% |
Growth driven by new patient populations |
Key Factors Impacting Sales
- Patent Status and Generics: Patent expiry anticipated around 2025-2026 could decrease prices by 40-60% and erode market share unless new formulations or indications are introduced.
- Regulatory Environment: Approval of biosimilars and generics may expand access but reduce profit margins.
- Market Penetration: Expansion into emerging markets (BRICS, Southeast Asia) presents growth opportunities; however, pricing and healthcare infrastructure limitations may slow adoption.
Pricing Dynamics
- Brand Name: Original branded formulations listed at approximately $2.50–$4.00 per tablet depending on strength.
- Generics: Prices fall to $0.50–$1.20 per tablet post-patent expiry.
- Reimbursement Trends: Insurance plans increasingly favor fixed-dose combinations, influencing sales positively in mature markets.
Regional Variations
- North America: Highest current sales, driven by extensive prescribing and well-established healthcare infrastructure.
- Europe: Similar trends, with some countries favoring monotherapies; growth depends on formulary inclusion.
- Asia-Pacific: Significant growth potential with expanding healthcare access and population size.
Strategic Recommendations
- Develop next-generation formulations with improved dosing or additional indications.
- Invest in marketing campaigns targeting physicians, especially in emerging markets.
- Prepare for patent expirations with cost-optimized manufacturing and diversified product pipelines.
- Engage with regulatory agencies for faster approvals and favorable positioning.
Key Takeaways
- Amlodipine/benazepril is positioned in a mature but growing segment of antihypertensive therapy.
- Sales are projected to grow at an approximately 12% CAGR from 2023 to 2028.
- Patent expirations around 2025-2026 may lead to price reductions and increased generic competition, potentially impacting margins.
- Market expansion into emerging regions offers substantial upside.
- Strategic innovation and regulatory navigation remain critical to maintaining growth.
FAQs
1. What factors influence the growth of amlodipine/benazepril sales?
Factors include hypertension prevalence, physician prescribing habits, formulary acceptance, patent status, and regional healthcare infrastructure.
2. How will patent expirations affect market dynamics?
Patent expirations around 2025-2026 could lead to significant price declines and increased generic competition, reducing revenue per unit.
3. Which regions present the most growth opportunities?
Emerging markets in Asia-Pacific, Latin America, and parts of Africa offer significant growth potential due to rising hypertension rates and expanding healthcare access.
4. How does pricing impact market penetration?
Lower prices through generics increase access and volume, especially in cost-sensitive regions, but may reduce profit margins for branded manufacturers.
5. What are future product development trends for combination antihypertensives?
Investments focus on formulations offering better adherence, novel agents with improved safety profiles, and multi-indication treatments.
References
[1] ReportLinker. “Global Antihypertensive Drugs Market Report 2022.”
[2] IQVIA Sales Data. “Hypertension Treatment Market Analysis.”
[3] FDA and EMA approval databases.
[4] Industry reports on patent expirations and generic market entry.