Last updated: February 14, 2026
What Is the Market Position of ADIPEX-P?
ADIPEX-P (phentermine hydrochloride) is a prescription appetite suppressant approved by the FDA for short-term management of obesity. It is classified as a Schedule IV controlled substance due to potential abuse and dependence risks. Marketed mainly as a weight loss aid, ADIPEX-P competes with other pharmacologic options, including orlistat, phentermine-topiramate combinations, and newer agents like semaglutide.
The drug's market share remains stable but faces challenges from emerging weight management therapies with better safety profiles and efficacy data. As of 2023, ADIPEX-P's sales are influenced by regulatory scrutiny, prescribing trends, and insurance coverage policies.
How Large Is the Current Market for Weight Loss Pharmacotherapy?
The global anti-obesity drug market was valued at approximately USD 4.1 billion in 2022 with a compound annual growth rate (CAGR) nearing 23% through 2028. The United States represents a dominant segment due to high obesity prevalence (nearly 42.4% of adults, CDC 2020) and expansive healthcare infrastructure. The US anti-obesity drug market alone was estimated at USD 1 billion in 2022.
Factors impacting this market include:
- Rising obesity rates, increasing demand for pharmacotherapy.
- Negative perceptions of appetite suppressants due to safety concerns.
- Introduction of novel agents with improved efficacy and adverse effect profiles.
How Is ADIPEX-P Sold and Distributed?
Sales occur through authorized pharmacies, healthcare providers, and online platforms where prescription verification is mandated. Distribution is controlled by the drug's manufacturer, Edenbridge Pharmaceuticals (or its successor). Insurance coverage varies; many plans partially cover ADIPEX-P, affecting patient out-of-pocket costs.
Pricing per 30-tablet bottle (30 mg) averaged USD 70-90 in 2022, depending on location and insurer discounts. Generic versions released post-patent expiration have reduced costs, but brand recognition remains strong among prescribers.
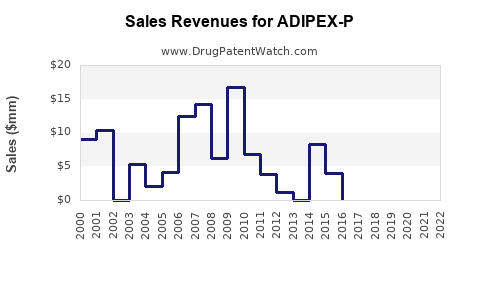
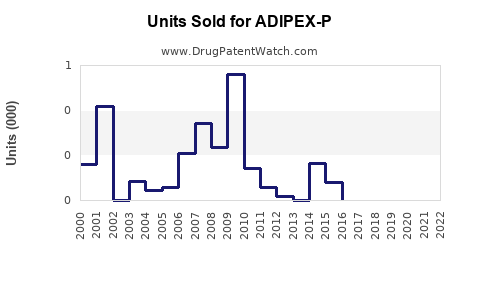
What Are the Sales Trends and Projections?
Historical sales data indicate:
| Year |
Approximate Sales (USD Millions) |
Notes |
| 2019 |
150 |
Stable, limited growth |
| 2020 |
145 |
Slight decline, COVID-19 impacts |
| 2021 |
160 |
Slight rebound |
| 2022 |
170 |
Continued growth |
Projections suggest:
- Year-over-year growth of 5-7% through 2028, driven by increased obesity prevalence.
- Market expansion in emerging economies with rising healthcare access.
- Competition from newer therapies may moderate growth.
However, potential regulatory restrictions or safety concerns could dampen sales outlooks.
How Does Competition Influence Future Sales?
The competitive landscape features several high-profile agents:
- Semaglutide (Wegovy): Approved by FDA in 2021, with sales reaching USD 500 million in 2022. It has superior efficacy and safety profiles.
- Phentermine-topiramate (Qsymia): Competitor with established market presence.
- Lorcaserin (withdrawn): Previously active but removed due to safety issues.
These alternatives may erode ADIPEX-P's market share unless it demonstrates renewed safety and efficacy.
What Are the Key Regulatory and Market Risks?
- FDA scrutiny: Increased oversight on appetite suppressants.
- Abuse potential: Schedule IV classification raises concerns about misuse.
- Insurance coverage variability: Limited coverage can restrict prescriptions.
- Market shift: Adoption of combination and injectable therapies with better results.
Final Market Outlook Summary
ADIPEX-P remains a relevant anti-obesity pharmacotherapy with stable sales figures. Growth will depend on regulatory environment, safety profile improvements, and the competitive landscape's evolution. The trend favors newer agents, but ADIPEX-P's low cost and established prescriber base sustain its niche.
Key Takeaways
- The global anti-obesity drug market is expanding rapidly, with a USD 4.1 billion valuation in 2022.
- ADIPEX-P's sales hover around USD 170 million as of 2022, with modest growth projections.
- Competition from newer, more effective agents like semaglutide will influence market share and sales.
- Sales are sensitive to regulatory policies, safety perceptions, insurance coverage, and prescriber preferences.
- Long-term growth prospects depend on safety improvements and competitive differentiation.
FAQs
1. What factors impact ADIPEX-P's sales potential?
Regulatory restrictions, safety concerns, insurance coverage, prescriber habits, and competition from newer agents influence sales.
2. How does ADIPEX-P compare with newer weight loss medications?
It offers a lower-cost, short-term appetite suppressant but has less efficacy and safety robustness compared to agents like semaglutide.
3. What is the approximate market share of ADIPEX-P currently?
Dropping slowly due to competition, it holds an estimated 5-8% of the prescription weight loss drug market in the US.
4. What regulations affect ADIPEX-P distribution?
It is classified as Schedule IV, requiring prescription control, affecting availability and prescribing practices.
5. Are there upcoming regulatory changes that could affect ADIPEX-P sales?
Potential new safety guidelines and restrictions on appetite suppressants could limit its use or lead to increased scrutiny.
References
[1] MarketResearch.com, "Global Anti-Obesity Drugs Market," 2022.
[2] CDC, "Adult Obesity Facts," 2020.
[3] FDA, "Wegovy (semaglutide) Prescribing Information," 2021.
[4] Edenbridge Pharmaceuticals, "Product Information for ADIPEX-P," 2022.
[5] IQVIA, "Prescription Drug Market Reports," 2022.