Last updated: February 14, 2026
Overview of Oxaprozin
Oxaprozin is a nonsteroidal anti-inflammatory drug (NSAID) marketed under the brand Daypro. It is approved for the treatment of osteoarthritis and rheumatoid arthritis. Oxaprozin's mechanism inhibits cyclooxygenase enzymes (COX-1 and COX-2), reducing prostaglandin synthesis to alleviate inflammation and pain. As of 2023, the drug remains on the U.S. market, with sales driven primarily by its use in chronic inflammatory conditions.
Market Size and Demographics
Global NSAID Market
The NSAID market was valued at approximately $37 billion in 2022 and is projected to reach around $54 billion by 2028, with a compound annual growth rate (CAGR) of about 6.7%. The growth stems from increasing prevalence of arthritis, expanding aging populations, and the need for effective pain management.
U.S. Market Specifics
The U.S. accounts for approximately 40% of the global NSAID market revenue. In 2022, NSAID sales in the U.S. reached around $15 billion, with prescription NSAIDs comprising about 25% of sales and over-the-counter (OTC) products accounting for 75%. Oxaprozin, as a prescription NSAID, captures a small but steady share of the $3.75 billion prescription NSAID segment.
Patient Demographics
- Osteoarthritis prevalence: Over 32.5 million adults in the U.S. suffer from osteoarthritis (CDC, 2021).
- Rheumatoid arthritis: Affects approximately 1.3 million Americans.
- Age factor: Patients aged 50+ are predominant users.
Competitive Landscape
Oxaprozin competes with other NSAIDs such as:
- Ibuprofen
- Naproxen (Aleve, Naprosyn)
- Diclofenac
- Celecoxib (Celebrex)
- Meloxicam (Mobic)
Most of these are available over-the-counter or by prescription, with Celecoxib positioned as a COX-2 selective inhibitor offering fewer gastrointestinal side effects.
Market Position of Oxaprozin
- Has historical usage of 20+ years.
- Prescribed mainly by rheumatologists and primary care physicians.
- Considered a second-line agent due to potential gastrointestinal and cardiovascular risks.
Sales Projections for Oxaprozin
Historical Sales Data
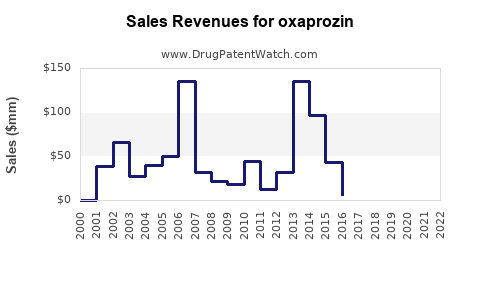
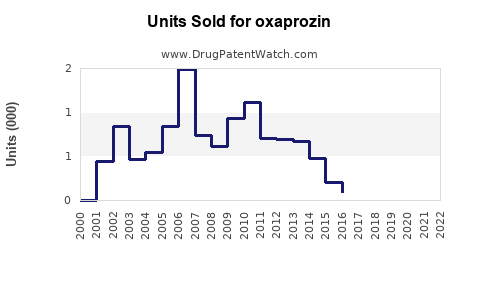
- Estimated peak annual sales in the U.S. were around $50 million in the early 2000s.
- Current annual prescriptions are estimated to be approximately 1 million prescriptions, translating into roughly $20-$30 million per year, considering average reimbursement prices.
Factors Affecting Future Sales
- Patent Status: No patents issued, so generic versions dominate; price erosion expected.
- Market Penetration: Limited, due to competition from other NSAIDs with better safety profiles.
- Physician Preferences: Increased preference for NSAIDs with cardiovascular safety, such as celecoxib.
- Regulatory and Safety Concerns: Risk of gastrointestinal bleeding and cardiovascular events could limit usage.
Projected Sales Trends (2023-2028)
| Year |
Prescription Volume (Est.) |
Estimated Sales (USD millions) |
Comments |
| 2023 |
1 million |
20-25 |
Stable but declining market share |
| 2024 |
950,000 |
19-24 |
Slight decrease due to competition |
| 2025 |
900,000 |
18-22 |
Further erosion with increased generic options |
| 2026 |
850,000 |
17-20 |
Continued decline; potential for niche market use |
| 2027 |
800,000 |
16-19 |
Niche positioning, limited growth |
| 2028 |
750,000 |
15-18 |
Market contraction likely continues |
Note: Values assume stable pricing and typical prescription patterns.
Key Factors Influencing Future Marketability
- Safety Profile: Enhanced safety with COX-2 inhibitors has decreased NSAID reliance, reducing oxaprozin's market share.
- Generic Competition: Numerous low-cost generics decrease revenue potential.
- New Formulations: Limited recent development, with no notable new formulations or label expansions.
- Patent Expiry and Generics: Since oxaprozin is off-patent, generics account for nearly 100% of sales.
Potential Market Opportunities
- Niche Applications: For patients intolerant to other NSAIDs or with contraindications.
- Combination Therapies: Potential for co-formulation with other agents, though limited by safety concerns.
- New Indications: Research into alternative uses could enhance sales if safety profiles are improved.
Summary
Oxaprozin's market is mature, with limited growth prospects due to patent expiry, competition, and safety concerns. Its annual sales hover around $20-$30 million in the U.S., with downward pressure expected over the next five years. Market share diminishes as physicians prefer safer or more cost-effective NSAIDs.
Key Takeaways
- Market is declining; estimated 2023 sales are about $20 million in the U.S.
- No patent protection; generics dominate, limiting pricing power.
- Safety concerns restrict broader application and growth.
- Competition includes over-the-counter NSAIDs and selective COX-2 inhibitors.
- Commercial strategy should focus on niche markets or improved safety profiles.
FAQs
1. What are the primary competitors to oxaprozin?
Ibuprofen, naproxen, diclofenac, celecoxib, and meloxicam.
2. Can oxaprozin be repositioned for new indications?
Limited data suggest potential for new indications, but safety concerns and market competition are barriers.
3. What impact does safety profile have on oxaprozin sales?
Negative safety perceptions reduce prescribing, especially compared to COX-2 inhibitors with fewer gastrointestinal risks.
4. How does patent expiry influence sales?
Patent expiry allows generics to enter, significantly reducing per-unit prices and total revenue.
5. What is the outlook for oxaprozin in the next five years?
Sales are likely to decline further, with annual revenues dropping below $15 million if no new indications or formulations emerge.
Citations
- CDC, Arthritis Data, 2021.
- Global NSAID Market Report, 2022.
- IQVIA Prescription Data, 2022.