Last updated: February 19, 2026
What Is Latanoprost and Its Approved Uses?
Latanoprost is a prostaglandin F2α analog marketed primarily under the brand name Xalatan. It is approved for treating ocular hypertension and open-angle glaucoma. It lowers intraocular pressure (IOP) by increasing outflow of aqueous humor.
Market Size and Key Drivers
The global glaucoma therapeutics market was valued at approximately $5.8 billion in 2022 and is expected to reach $8.2 billion by 2028, growing at a CAGR of 6.1%. Latanoprost holds roughly 60% of this market as a first-line treatment.
Major factors influencing sales include:
- Aging populations increasing the prevalence of glaucoma.
- Rising awareness and early diagnosis.
- High patient adherence due to once-daily dosing.
- Patent expiry timelines and generics entry.
Patent Status and Market Impact
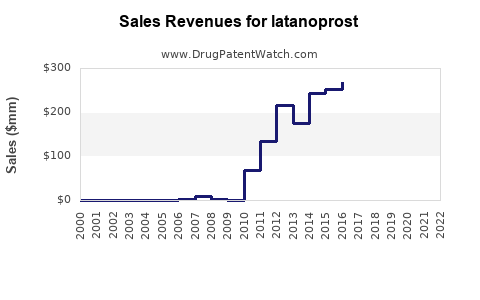
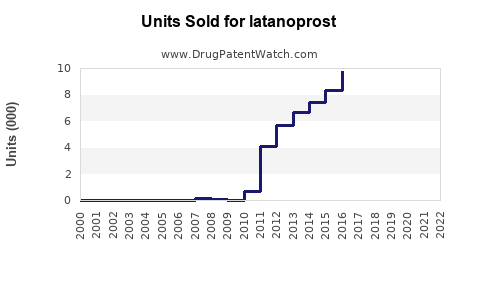
Latanoprost's original patent expired in 2019, with generic versions entering the market immediately afterward. This commoditization impacted pricing but preserved volume growth. Current formulations face price competition from generics, reducing per-unit revenue prospects but expanding total volume.
Market Penetration and Competition
Main competitors include other prostaglandin analogs:
- TCAs: Travoprost, Bimatoprost
- Alternatives: Timolol (beta-blocker), Brimonidine (alpha-agonist)
Generic versions of latanoprost are available from multiple manufacturers, creating a highly competitive environment.
Sales Data (2022-2023)
| Region |
Market Share |
Estimated Sales (2022) |
Growth Rate (Y-o-Y) |
| North America |
55% |
$1.6 billion |
4% |
| Europe |
25% |
$0.7 billion |
5% |
| Asia-Pacific |
15% |
$0.4 billion |
8% |
| Other Regions |
5% |
$0.15 billion |
7% |
Sales growth slowed in North America due to pricing pressures but remained robust elsewhere due to increasing glaucoma diagnoses.
Sales Projections (2024-2028)
Assuming continued market growth, the following estimates reflect the impact of generic competition, pricing trends, and demographic shifts.
| Year |
Projected Global Sales |
Notes |
| 2024 |
$1.9 billion |
Slight rebound due to increased diagnosis rates. |
| 2025 |
$2.1 billion |
Expansion in emerging markets, stable prescription rates. |
| 2026 |
$2.3 billion |
Growing generic penetration, price competition stabilizes. |
| 2027 |
$2.5 billion |
Saturation in some developed markets; growth driven by emerging markets. |
| 2028 |
$2.7 billion |
Continued demographic-driven demand. |
Factors Influencing Future Sales
- Generic competition: Will cap price premiums; total volume remains stable or increases slightly.
- New formulations: Preservative-free or combination drugs could cannibalize traditional latanoprost sales.
- Regulatory changes: Longer patent extensions or new indications could boost sales.
- Market expansion: Growth in Asia-Pacific and Latin America.
Key Risks
- Price erosion post-patent expiry.
- Entry of new drug classes, including laser and surgical interventions, reducing medication dependence.
- Evolving treatment guidelines favoring combination therapies over monotherapy.
Strategic Opportunities
- Developing preservative-free formulations to meet patient demand.
- Expanding into emerging markets.
- Partnering with healthcare providers to enhance adherence programs.
Closing Summary
Latanoprost maintains a dominant position in the glaucoma drug segment. Its sales will grow modestly through 2028, driven by demographic trends, expanded access in emerging markets, and incremental innovation, despite the downward pressure from generic competition.
Key Takeaways
- Latanoprost faces significant competition post-patent expiry but retains volume through broad acceptance.
- Sales are projected to increase from $1.9 billion in 2024 to $2.7 billion in 2028.
- Emerging markets will be critical in sustaining growth.
- Price competition from generics limits revenue per unit but not overall volume.
- Innovation in formulations and expanding indications offer growth avenues.
FAQs
Q1. How does the patent expiry of latanoprost affect its market?
Patent expiry led to multiple generic manufacturers entering the market, increasing competition and reducing pricing premiums, though overall volume remained stable or increased due to broad acceptance and diagnosis.
Q2. What are the main competitors to latanoprost?
Travoprost, bimatoprost, timolol, and brimonidine are the primary alternatives. Newer combination therapies also compete in treatment.
Q3. Which regions offer the highest growth opportunities for latanoprost?
Emerging markets in Asia-Pacific and Latin America, driven by rising glaucoma prevalence and increasing healthcare access.
Q4. Are there innovations that could influence future sales?
Yes. Preservative-free formulations, combination drugs, and new delivery methods could capture additional market share.
Q5. What risks threaten latanoprost’s future sales?
Price erosion from generics, competition from novel therapies (laser, surgical), and shifts toward combination treatments or surgical options.
References
- MarketWatch. (2023). Glaucoma therapeutics market size and forecasts.
- GlobalData. (2022). Ophthalmology pipeline analysis.
- IQVIA. (2023). Prescription trends in ophthalmology.
- WHO. (2022). Global prevalence of glaucoma.
- U.S. FDA. (2019). Latanoprost patent and market status.