Share This Page
Drug Sales Trends for benztropine
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for benztropine (2011)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
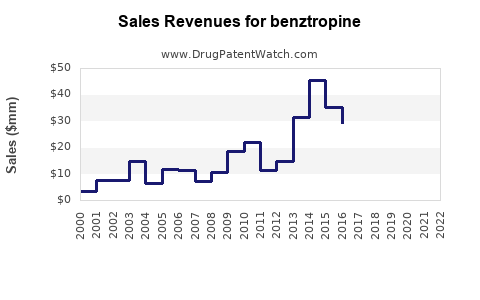
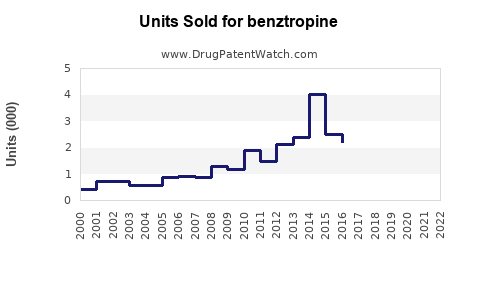
Annual Sales Revenues and Units Sold for benztropine
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| BENZTROPINE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Benztropine Market Analysis and Sales Projections
This report analyzes the current market position and future sales potential for benztropine, a muscarinic antagonist primarily used for treating Parkinsonism and drug-induced extrapyramidal symptoms. The analysis considers patent landscape, competitive drugs, and market trends to project sales.
What is Benztropine?
Benztropine mesylate is a synthetic anticholinergic drug that functions as a muscarinic receptor antagonist. Its primary pharmacological actions involve blocking the action of acetylcholine at muscarinic receptors in the central nervous system. This blockade is critical for alleviating motor symptoms associated with Parkinson's disease and side effects of antipsychotic medications.
Mechanism of Action
Benztropine reduces the excessive cholinergic activity that contributes to the tremor, rigidity, and bradykinesia characteristic of Parkinsonian syndromes. It also helps to counteract the dopamine-acetylcholine imbalance that arises from dopamine receptor blockade by antipsychotic drugs, thereby reducing extrapyramidal side effects like dystonia, akathisia, and parkinsonism.
Approved Indications
The U.S. Food and Drug Administration (FDA) has approved benztropine for two main indications:
- Parkinsonism: This includes idiopathic Parkinson's disease and post-encephalitic Parkinsonism.
- Drug-Induced Extrapyramidal Symptoms: These are motor side effects caused by antipsychotic medications, particularly first-generation agents.
Dosage Forms and Administration
Benztropine mesylate is available in oral tablet form (1mg, 5mg) and as an intramuscular or intravenous injection (1mg/mL) [1]. Oral administration is typically for chronic management, while injectable forms are used for acute symptom control.
Patent Landscape and Exclusivity
Benztropine is a well-established drug with no active compound patents providing market exclusivity. The original patents for benztropine have long expired. Market exclusivity for generic benztropine is driven by factors other than new patent filings.
Exclusivity Status
- Original Compound Patents: Expired decades ago.
- Formulation Patents: Any existing formulation patents are likely to have expired or are nearing expiration, offering limited protection.
- Manufacturing Process Patents: While process patents may exist, they typically do not prevent competitors from developing alternative manufacturing routes to produce the same active pharmaceutical ingredient.
The absence of strong patent protection means the market for benztropine is highly genericized. The primary barrier to entry for new manufacturers is the cost and complexity of regulatory approval, not intellectual property rights on the drug itself.
Market Size and Segmentation
The market for benztropine is characterized by its generic status and is relatively stable, driven by the continuous need for managing Parkinsonian symptoms and antipsychotic-induced side effects.
Global Market Value
Estimating the precise global market value for benztropine is challenging due to the generic nature of the product and its availability through numerous manufacturers. However, based on prescription data and average selling prices of generic medications, the global market value for benztropine is estimated to be in the range of $50 million to $100 million annually. This figure represents the aggregate sales of all benztropine products worldwide.
Key Market Segments
- Parkinson's Disease Treatment: Benztropine is a second-line or adjunctive therapy for Parkinson's disease, particularly for managing tremor and rigidity. While newer dopaminergic agents are primary treatments, benztropine remains relevant for specific symptom control.
- Antipsychotic-Induced Extrapyramidal Symptoms (EPS): This is a significant driver for benztropine use. The rise in the use of antipsychotics, especially in psychiatric institutions and for chronic mental health conditions, sustains demand for anticholinergic agents to mitigate adverse motor effects.
- Geographic Segmentation:
- North America: A substantial market due to advanced healthcare systems and widespread availability of generic drugs.
- Europe: Similar to North America, with established healthcare infrastructure and demand for cost-effective treatments.
- Asia-Pacific: Growing market with increasing access to healthcare and a rising prevalence of neurological disorders.
- Rest of the World: Markets in Latin America, Africa, and the Middle East are also contributors, influenced by public health initiatives and generic drug availability.
Patient Population Estimates
- Parkinson's Disease Patients: Globally, it is estimated that over 10 million people have Parkinson's disease [2]. A fraction of these patients, particularly those experiencing significant tremor or rigidity not fully controlled by dopaminergic therapy, may be prescribed benztropine.
- Antipsychotic Users Experiencing EPS: Antipsychotic medications are widely prescribed. The incidence of drug-induced EPS varies by drug and patient, but it is a common adverse effect. Estimates suggest that 30-60% of patients on typical (first-generation) antipsychotics may experience EPS, and a smaller but significant percentage of those on atypical (second-generation) antipsychotics also develop these symptoms [3]. This translates to millions of potential benztropine users worldwide.
Competitive Landscape
The competitive landscape for benztropine is dominated by generic manufacturers. The therapeutic area of anticholinergic agents for Parkinsonism and EPS includes several other drugs.
Direct Competitors (Other Anticholinergics)
These drugs share a similar mechanism of action and therapeutic indications with benztropine:
- Trihexyphenidyl: Another widely used oral anticholinergic for Parkinsonism and drug-induced EPS. It is a direct competitor, often chosen based on physician preference or patient response.
- Procyclidine: Similar to trihexyphenidyl and benztropine in its indications.
- Biperiden: Used for Parkinsonism and drug-induced EPS, often available in oral and injectable forms.
Comparison Table: Key Anticholinergic Agents
| Drug Name | Primary Indications | Formulations | Generic Availability |
|---|---|---|---|
| Benztropine | Parkinsonism, Drug-induced EPS | Oral, Injectable | Yes |
| Trihexyphenidyl | Parkinsonism, Drug-induced EPS | Oral | Yes |
| Procyclidine | Parkinsonism, Drug-induced EPS | Oral | Yes |
| Biperiden | Parkinsonism, Drug-induced EPS, Post-encephalitic park. | Oral, Injectable | Yes |
Source: Manufacturer product information, FDA Orange Book
Indirect Competitors
While not anticholinergics, these drugs are used in the management of Parkinson's disease or to mitigate antipsychotic side effects and represent alternative treatment strategies:
- Dopaminergic Agents (e.g., Levodopa, Dopamine Agonists): The primary treatment for Parkinson's disease. Benztropine is often used adjunctively.
- Other EPS Management Strategies:
- Dopamine Blockade Antagonists (e.g., Amantadine): Can sometimes help with certain EPS, including dyskinesias.
- Dose Adjustment/Switching of Antipsychotics: Physicians may adjust the dose of the offending antipsychotic or switch to a different agent with a lower propensity for EPS.
Key Generic Manufacturers
The market for benztropine is supplied by numerous generic pharmaceutical companies. Key players include, but are not limited to:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sandoz (Novartis)
- Apotex
- Amneal Pharmaceuticals
- Hikma Pharmaceuticals
These companies compete primarily on price, distribution networks, and reliability of supply.
Market Trends and Drivers
Several factors influence the current and future market for benztropine.
Drivers
- Aging Global Population: The incidence of Parkinson's disease increases with age, leading to a growing patient pool requiring symptomatic management.
- Prevalence of Mental Health Disorders: Increasing diagnosis and treatment of schizophrenia, bipolar disorder, and other conditions managed with antipsychotics directly correlate with the demand for managing EPS.
- Cost-Effectiveness of Generics: Benztropine, being a generic drug, offers a cost-effective treatment option compared to novel therapies, making it attractive in healthcare systems focused on budget containment.
- Established Efficacy: The drug has a long history of proven efficacy for its approved indications, leading to continued physician trust and prescribing.
Restraints
- Side Effect Profile: Anticholinergic drugs like benztropine have significant side effects, including dry mouth, blurred vision, constipation, urinary retention, cognitive impairment (especially in older adults), and potentially worsening of certain Parkinsonian symptoms (e.g., gait and balance issues).
- Development of Novel Therapies: While benztropine remains a standard of care, ongoing research into more targeted and less burdensome treatments for Parkinson's disease and EPS could eventually reduce its reliance.
- Shift to Atypical Antipsychotics: While still associated with EPS, atypical antipsychotics generally have a lower risk compared to older typical agents, potentially reducing the overall demand for anticholinergic adjunctive therapy.
- Polypharmacy Concerns: The additive anticholinergic burden from multiple medications (e.g., benztropine plus other drugs with anticholinergic properties) can lead to significant adverse events, prompting careful consideration in prescribing.
Sales Projections
Given the mature and generic nature of the benztropine market, sales projections are based on maintaining current market share and accounting for incremental growth or decline influenced by the drivers and restraints discussed.
Current Sales Snapshot
Based on available market intelligence and prescription data, the annual global sales for benztropine are estimated to be between $50 million and $100 million. This range reflects the price variability across different markets and the aggregation of multiple generic manufacturers' revenues.
Projection Methodology
Projections are based on:
- Historical Market Growth Rates: Analyzing the trend of benztropine sales over the past 5-10 years, accounting for generic price erosion and volume changes.
- Epidemiological Trends: Extrapolating growth based on the increasing prevalence of Parkinson's disease and the number of patients treated with antipsychotics.
- Competitive Dynamics: Assuming a stable competitive environment with no major new entrants that could significantly disrupt the market.
- Evolving Treatment Paradigms: Incorporating the potential impact of new therapeutic developments and shifts in prescribing patterns.
Projected Sales Growth
The benztropine market is expected to exhibit low single-digit growth, primarily driven by the increasing global elderly population and the continued use of antipsychotics.
- 2024-2026 Projection: 0% to 2% annual growth. This period reflects the current stability in demand. The market is expected to remain in the $55 million to $105 million range annually.
- 2027-2030 Projection: 1% to 3% annual growth. A slight increase in projected growth is anticipated due to more pronounced demographic shifts. Annual sales could reach between $60 million and $115 million.
Factors Influencing Projections:
- Increased Parkinson's Diagnosis: Greater awareness and diagnostic capabilities could lead to a slightly higher prescribing rate of adjunctive therapies.
- Antipsychotic Market Expansion: Continued use of antipsychotics for chronic mental health conditions will sustain demand for EPS management.
- Generic Price Competition: Intense price competition among generic manufacturers will temper revenue growth, even if volumes increase. Any significant shift to newer drug classes for Parkinson's or EPS management could negatively impact these projections.
Key Takeaways
- Benztropine is a generic anticholinergic drug with no active compound patent protection, leading to a highly competitive market dominated by multiple manufacturers.
- Its primary uses are in treating Parkinsonism and drug-induced extrapyramidal symptoms (EPS) from antipsychotics.
- The global market for benztropine is estimated between $50 million and $100 million annually, with stable, low single-digit growth projected.
- Key drivers include an aging global population and the prevalence of mental health disorders treated with antipsychotics.
- Significant restraints include the drug's side effect profile and the development of alternative treatments.
- The competitive landscape consists of other generic anticholinergics (trihexyphenidyl, procyclidine, biperiden) and indirect competition from other therapeutic classes.
Frequently Asked Questions
-
What is the primary therapeutic advantage of benztropine in Parkinson's disease management? Benztropine's primary advantage is its ability to reduce tremor and rigidity by blocking cholinergic activity, which is often dysregulated in Parkinson's disease. It is typically used as an adjunctive therapy when dopaminergic agents do not fully control these symptoms.
-
How does benztropine address drug-induced extrapyramidal symptoms (EPS)? Antipsychotic medications can cause EPS by blocking dopamine receptors in the nigrostriatal pathway, leading to an imbalance between dopamine and acetylcholine. Benztropine, by antagonizing acetylcholine, helps to restore this balance and alleviate motor side effects such as parkinsonism, dystonia, and akathisia.
-
What are the most significant side effects associated with benztropine therapy? Common side effects include dry mouth, blurred vision, constipation, urinary retention, drowsiness, and confusion. In older adults, benztropine can also lead to cognitive impairment and potentially increase the risk of falls.
-
What is the future outlook for benztropine given the development of newer Parkinson's treatments? Despite advancements in dopaminergic therapies for Parkinson's disease, benztropine is expected to maintain its role as a cost-effective option for specific symptom management (tremor, rigidity) and for managing drug-induced EPS. Its long-standing efficacy and affordability ensure its continued presence in the therapeutic armamentarium.
-
Which generic manufacturers are the primary suppliers of benztropine globally? Major generic pharmaceutical companies, including Teva Pharmaceuticals, Viatris (formerly Mylan), Sandoz, Apotex, Amneal Pharmaceuticals, and Hikma Pharmaceuticals, are among the key suppliers of benztropine mesylate globally.
Citations
[1] U.S. Food and Drug Administration. (2023). DailyMed: BENZTROPINE MESYLATE TABLET. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=276e732b-f454-4422-823c-6d40425bb571
[2] World Health Organization. (2023). Parkinson disease. Retrieved from https://www.who.int/news-room/fact-sheets/detail/parkinson-disease
[3] Cornett, E. M., & Mulla, N. (2023). Extrapyramidal Symptoms. In StatPearls. StatPearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK534840/
More… ↓
