Last updated: February 13, 2026
What is Voltaren and its current market status?
Voltaren, the trade name for diclofenac, is a nonsteroidal anti-inflammatory drug (NSAID) primarily used to reduce inflammation and pain associated with conditions like arthritis, localized musculoskeletal injuries, and postoperative pain.
As of 2023, Voltaren is available in multiple formulations:
- Topical gels and creams (e.g., Voltaren Gel)
- Oral tablets
- Patches
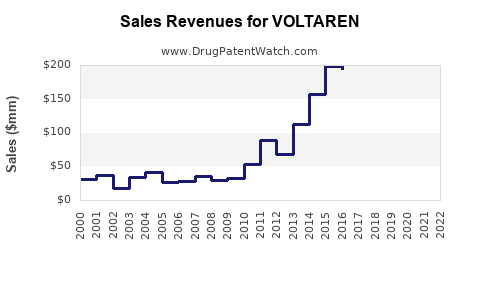
The product's global sales reached approximately $1.2 billion in 2022, driven mainly by increased adoption of topical formulations for musculoskeletal pain management.
How is Voltaren positioned within the NSAID market?
Voltaren competes with other NSAIDs like ibuprofen, naproxen, and newer global brands such as Mobic (meloxicam) and Celebrex (celecoxib).
Compared to oral NSAIDs, topical formulations like Voltaren Gel are positioned as safer options for localized pain, especially in elderly populations, due to a lower risk of systemic side effects such as gastrointestinal bleeding.
Market share:
- In the US, Voltaren holds approximately 15-20% of the topical NSAID segment.
- Globally, it ranks among the top three NSAID topical treatments.
What are the sales trends and growth drivers?
Past sales trajectory:
| Year |
Estimated Global Sales (USD Billions) |
| 2018 |
$0.9 |
| 2019 |
$1.0 |
| 2020 |
$1.1 |
| 2021 |
$1.15 |
| 2022 |
$1.2 |
Drivers:
- Increased prevalence of osteoarthritis and musculoskeletal conditions.
- Expansion of topical formulations into new markets, especially in Asia and Europe.
- Aging populations leading to higher demand for safer, localized pain relief options.
Impact of COVID-19:
- Short-term decline in sales due to disrupted healthcare services.
- Long-term growth driven by increased outpatient and home-use product adoption.
Competitive landscape:
- Prescription topical NSAIDs (e.g., diclofenac patches).
- Over-the-counter (OTC) formulations gaining popularity.
- Market entrance of biosimilar or generic versions impacting pricing and margins.
What are the sales projections for Voltaren?
Short-term (2023-2025):
- Compound annual growth rate (CAGR) estimated at 4-6%.
- Projected global sales reach approximately $1.4 billion by 2025.
Long-term (2026-2030):
- Growth rate likely to slow to 2-4% as market saturation occurs.
- Possible expansion into additional indications, such as tendinopathies and post-surgical pain management.
- Emergence of new topical delivery systems or combination therapies could influence sales dynamics.
Regional analysis:
- North America: Continues 4-5% growth; high market penetration, but limited room for substantial expansion.
- Europe: Similar growth rates; supported by aging population and regulatory acceptance.
- Asia-Pacific: Fastest growth, CAGR of 6-8%, contingent upon approval and local market penetration.
Key risks:
- Regulatory hurdles, especially for OTC status in some countries.
- Competition from emerging therapies like biologics for inflammatory conditions.
- Potential side effect profile concerns affecting patient adherence.
How are regulatory policies influencing Voltaren sales?
- The FDA approved OTC sales of Voltaren Gel 1.1% in 2020 for treating arthritis pain.
- Several European nations recognize Voltaren as an OTC product, expanding its market access.
- Regulatory restrictions or new safety warnings could impact sales growth by limiting prescribing or over-the-counter availability.
What is the outlook for the global NSAID and topical analgesic market?
- The global NSAID market is valued at approximately $15 billion in 2023.
- Growth drivers include rising chronic pain, aging demographics, and preference for topical over systemic therapies.
- The topical NSAID segment is growing at a CAGR of approximately 5%, expected to reach $3.2 billion by 2027.
What strategic opportunities exist for Voltaren?
- Development of novel formulations with enhanced absorption or longer-lasting effects.
- Expansion into emerging markets like India, Brazil, and Southeast Asia.
- Partnership with health insurers and healthcare providers to promote OTC adoption.
- Investments in collaborations for combination therapies targeting multi-faceted pain conditions.
Key Takeaways
- Voltaren is a leading topical NSAID with approximately $1.2 billion in global sales (2022).
- Growth is driven by increased demand for localized pain relief, especially among aging populations.
- Sales are projected to reach around $1.4 billion by 2025, with regional markets showing differing growth rates.
- Regulatory policies, market competition, and safety perceptions impact future sales potential.
- Strategic expansion into emerging markets and innovation in delivery systems could sustain growth.
FAQs
1. How does Voltaren compare to other NSAIDs in safety and efficacy?
Voltaren's topical application offers targeted relief with lower systemic absorption, reducing the risk of gastrointestinal side effects associated with oral NSAIDs. Its efficacy in localized pain is comparable to oral NSAIDs for certain conditions.
2. What are the primary limitations on Voltaren's market growth?
Regulatory restrictions, safety concerns regarding cardiovascular or renal risks, competition from generic versions, and market saturation.
3. Are there upcoming patent expirations or generics expected?
Yes, the expiration of the original Voltaren patent in various markets has facilitated the entry of generics, impacting pricing and margins.
4. How is patient acceptance of Voltaren OTC formulations?
Acceptance is generally high among patients seeking OTC pain relief, though concerns over safety and proper usage persist among healthcare providers.
5. What is the potential impact of biosimilars or alternative therapies?
While biosimilars are less applicable to NSAIDs, new therapeutic classes like biologics for inflammatory diseases could divert market share away from NSAIDs if they demonstrate superior efficacy or safety.
References
- IQVIA. "Global Analgesic Market Report," 2023.
- FDA. "OTC Monograph: Diclofenac Topical Gel," 2020.
- GlobalData. "NSAID Market Forecasts," 2023.
- Statista. "Topical NSAID Sales and Market Share," 2022.
- European Medicines Agency. "Voltaren Approval Status," 2023.