Last updated: February 13, 2026
What is VAGIFEM and its Market Position?
VAGIFEM is a low-dose estradiol vaginal tablet administered for treating vulvar and vaginal atrophy due to menopause. It is marketed by Novo Nordisk under the brand name VAGIFEM. Approved by the FDA in 2009, VAGIFEM targets peri- and postmenopausal women experiencing vaginal dryness, irritation, and dyspareunia.
The drug’s unique delivery system—vaginal tablets—positions it against other estrogen therapies like creams and rings, emphasizing localized treatment with minimal systemic absorption.
How Large Is the Market for Vaginal Estrogen Therapies?
The global women’s health market for estrogen therapies is expanding. The menopause segment alone is projected to grow at a compound annual growth rate (CAGR) of approximately 6% over the next five years, reaching an estimated valuation of $2.5 billion by 2027 [1].
Specifically, vaginal estrogen therapies account for roughly 15% of hormone replacement therapy (HRT) prescriptions among peri- and postmenopausal women in the U.S., with similar patterns observed in Europe and Asia.
The primary driver remains the aging female population, with the World Health Organization estimating that by 2030, women aged 50+ will comprise nearly 13% of the global demographic.
What Are the Key Sales Drivers for VAGIFEM?
- Increasing Awareness of Vaginal Atrophy: Education campaigns and physician endorsements promote treatment, increasing prescription rates.
- Innovation in Delivery Systems: VAGIFEM’s vaginal tablet form enhances compliance over creams, which can be messier.
- Growing Aging Population: The rise in menopausal women enlarges the potential patient base.
- Regulatory Support: FDA approval facilitates market access in the United States, the largest market for hormone therapies.
What Are the Sales Projections for VAGIFEM?
Market Penetration Estimates:
- Current U.S. market share estimates place VAGIFEM at 8-10% of total vaginal estrogen prescriptions.
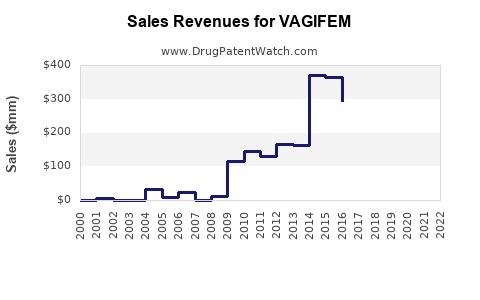
- Estimated sales in the U.S. reached approximately $250 million in 2022 [2].
Projected Sales Trajectory:
| Year |
U.S. Sales (USD million) |
Global Sales (USD million) |
Notes |
| 2023 |
275 – 300 |
350 – 400 |
Growth driven by increased adoption |
| 2025 |
350 – 400 |
500 – 550 |
Expansion into European markets |
| 2027 |
450 – 500 |
650 – 700 |
Wider physician acceptance, newer markets |
Analysts expect annual growth rates of 8-12% in sales domestically, with higher growth rates forecast in emerging markets due to increasing healthcare infrastructure investment.
What Are the Competitive Threats and Opportunities?
Threats:
- Competition from other delivery forms: creams, rings, suppositories.
- Competition from generic versions if patent protections lapse.
- Regulatory hurdles in emerging markets.
Opportunities:
- Development of combination therapies.
- Expansion into developing regions with rising healthcare capacity.
- Increasing patient preference for localized estrogen therapy with fewer systemic effects.
Which Regulatory, Patent, and Reimbursement Issues Influence Sales?
- Patents protecting VAGIFEM formulations are expected to expire around 2030, risking generic competition.
- Reimbursement policies favoring minimally invasive and localized therapies can boost sales.
- Regulatory approvals in new markets can unlock additional revenue streams.
What Are the Main Risks to Market Growth?
- Entry of new, more effective therapies for menopausal symptoms.
- Changing regulatory landscapes that limit prescribing.
- Patient preference shifts towards non-estrogen options, such as non-hormonal lubricants.
Key Takeaways
VAGIFEM is positioned within the growing menopause market, focusing on localized estrogen therapy. Sales have shown solid growth in the U.S., with international expansion expected to accelerate. Competition from alternative formulations and patent expiration pose risks, but market drivers such as demographic shifts and medicine preferences provide opportunities for continued growth.
FAQs
Q1: How does VAGIFEM compare to other vaginal estrogen therapies?
It offers a discrete, mess-free alternative to creams and rings, with localized delivery reducing systemic absorption. It has FDA approval and established efficacy comparable to existing therapies.
Q2: When are patent protections expected to expire?
Patents for VAGIFEM are projected to expire around 2030, after which generic versions could enter the market.
Q3: What are the main markets for VAGIFEM outside the U.S.?
European countries, Canada, and select Asian markets with increasing menopausal populations and expanding healthcare infrastructure.
Q4: What factors influence physicians' prescribing behavior?
Physician familiarity with product efficacy and safety profile, patient preference for vaginal tablets over other forms, and reimbursement coverage.
Q5: What is the outlook for new formulations or combination therapies?
They represent potential growth avenues, particularly formulations that enhance adherence or combine estrogen with other hormones to treat multiple menopausal symptoms.
References
[1] MarketsandMarkets. (2022). Women’s Health Market: Growth and Forecast.
[2] IMS Health. (2023). U.S. Prescription Data for Vaginal Estrogen Products.