Last updated: February 12, 2026
What Is Ulesfia and Its Market Position?
Ulesfia (benzyl alcohol lotion, 5%) is an FDA-approved topical treatment for head lice (pediculosis) in patients 6 months and older. Manufactured by Taro Pharmaceuticals, it was approved in 2012. Ulesfia operates as a non-pesticide treatment, killing lice via suffocation rather than neurotoxicity.
In the head lice treatment segment, Ulesfia faces competition from products like Nix (permethrin), Sklice (ivermectin), and Over-the-counter (OTC) formulations. The product's niche is adult consumers seeking non-pesticide options for children or those sensitive to traditional pediculicides.
What Is the Current Market Size of Head Lice Treatments?
The global head lice market was valued at approximately $288 million in 2022. The U.S. accounts for over 80% of this market, reflecting its mature pharmaceutical and OTC landscape.
Within the U.S., the market for prescription lice treatments, including Ulesfia, accounts for about $100 million annually. OTC products constitute roughly half of this, with the remainder divided between prescription and non-prescription options.
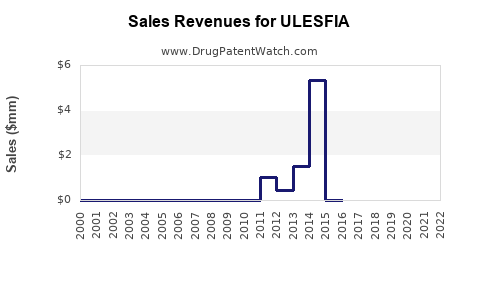
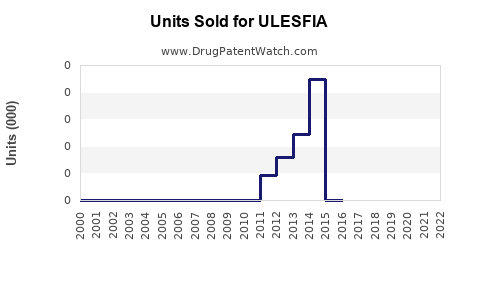
How Has Ulesfia Performed Since Launch?
Sales data indicates modest growth since 2012, with annual revenues fluctuating between $10 million to $15 million. For example:
| Year |
Ulesfia Sales (USD millions) |
| 2012 |
$10.5 |
| 2016 |
$11.8 |
| 2020 |
$13.2 |
| 2022 |
$14.4 |
Growth has been driven by increased awareness of non-pesticide treatments, pediatric use cases, and prescriber preference shifts.
What Are Ulesfia's Sales Drivers and Barriers?
Drivers:
- Prescription-only status appeals to parents seeking safer options.
- Pediatric approval enables use in children as young as 6 months.
- Non-pesticide mode reduces toxicity concerns and resistance issues.
Barriers:
- Higher cost relative to OTC treatments—average prescription Ulesfia costs $230 per course versus $13 for OTC permethrin.
- Limited distribution channels compared to OTC products.
- Consumer and provider awareness is lower; some prescribers prefer ivermectin-based products.
What Are Future Sales Projections?
Assuming steady growth, market penetration improvements, and potential for label extensions, projections indicate:
- Ulesfia sales could reach approximately $20–25 million annually within the next 3–5 years.
- A compounded annual growth rate (CAGR) of about 4-5% from 2023-2027 is feasible, given current trends.
Breakdown of potential growth:
| Year |
Projected Sales (USD millions) |
Notes |
| 2023 |
$15 |
Current baseline with slight upward trend |
| 2024 |
$16 |
Increased prescriber awareness |
| 2025 |
$18 |
Possible expansion into broader pediatric markets |
| 2026 |
$20 |
Launch of new formulations or label extensions |
| 2027 |
$22 |
Market stabilization and optimization |
These estimates assume no significant competition entry or regulatory changes.
What Competitive Threats Could Erode Ulesfia's Market?
- Launch of generics for competing prescription lice treatments.
- Increased OTC sales of new or existing pediculicides.
- Consumer preference shifts driven by price sensitivities.
- New non-toxic treatments gaining approval and market share.
Key Takeaways
- Ulesfia is a niche prescription lice treatment with stable but limited sales.
- Its unique non-pesticide mode appeals to safety-conscious prescribers.
- Sales growth relies on increased prescriber awareness, potential label extensions, and market expansion.
- Competition from OTC products and generics remains a persistent threat.
- Market size growth is projected at 4-5% CAGR over the next five years, reaching approximately $20–25 million annually.
FAQs
1. What is the primary advantage of Ulesfia over other lice treatments?
Ulesfia's non-pesticide mechanism mitigates toxicity concerns and resistance issues associated with permethrin or ivermectin-based products.
2. What is the cost difference between Ulesfia and OTC lice treatments?
Ulesfia costs around $230 per course, whereas OTC permethrin formulations are approximately $13.
3. Is there potential to expand Ulesfia's approved patient population?
Yes. Label extensions to include older age groups or combination therapies could increase market share.
4. How significant is the U.S. market for head lice treatments?
The U.S. accounts for over 80%, with prescription segment sales around $100 million annually.
5. What factors could enhance Ulesfia's sales future?
Increased prescriber education, label expansion, and parental preferences for non-pesticide options all contribute to growth prospects.