Share This Page
Drug Sales Trends for TOPIRAMATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TOPIRAMATE (2011)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
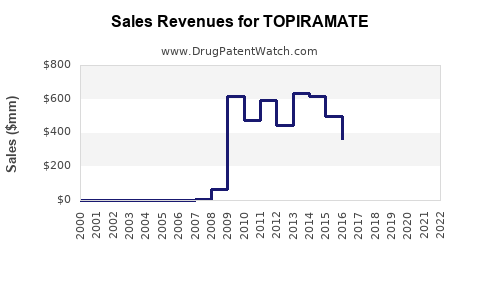
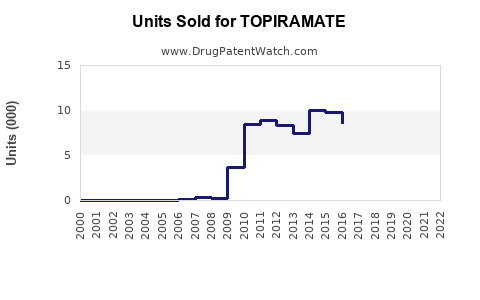
Annual Sales Revenues and Units Sold for TOPIRAMATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TOPIRAMATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TOPIRAMATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TOPIRAMATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TOPIRAMATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Topiramate Market Analysis and Sales Projections
This report analyzes the current market landscape and forecasts sales for topiramate, a broad-spectrum antiepileptic and migraine prophylactic drug. The analysis covers patent expirations, generic competition, therapeutic indications, and market growth drivers, concluding with projected sales figures through 2030.
What is Topiramate and What are Its Primary Indications?
Topiramate is an anticonvulsant and anti-migraine medication. Its mechanism of action is multifaceted, involving blockade of voltage-gated sodium channels, enhancement of gamma-aminobutyric acid (GABA) neurotransmission, antagonism of AMPA/kainate receptors, and inhibition of carbonic anhydrase [1].
Primary indications for topiramate include:
- Epilepsy: Specifically for partial-onset seizures and generalized tonic-clonic seizures in adults and children [2].
- Migraine Prophylaxis: Prevention of migraine headaches in adults and adolescents [2].
- Off-Label Uses: While not FDA-approved for these indications, topiramate is frequently prescribed off-label for conditions such as binge-eating disorder, bulimia nervosa, and weight management [3].
What is the Patent Status of Topiramate?
Topiramate was first patented by Johnson & Johnson (Ortho-McNeil Pharmaceutical) under U.S. Patent No. 4,513,001, which was granted on April 23, 1985, and expired in 2004 [4]. Subsequent patents related to formulations and methods of use have also expired or are nearing expiration.
Key patent milestones include:
- Original Composition of Matter Patent: Expired in 2004.
- Formulation Patents: Various patents covering specific dosage forms and delivery mechanisms have expired or are in the public domain.
- Method of Use Patents: Patents related to specific therapeutic applications, such as migraine prophylaxis, have also expired.
The expiration of primary patents has allowed for widespread generic entry, significantly impacting market dynamics.
Who are the Major Generic Manufacturers of Topiramate?
The market for topiramate is dominated by generic manufacturers following the expiry of its core patents. Major players include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Lupin Ltd.
- Alembic Pharmaceuticals Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
- Accord Healthcare Ltd.
These companies produce topiramate in various strengths and dosage forms, primarily oral tablets and capsules.
How is Topiramate Marketed and Distributed?
Topiramate is primarily distributed through retail pharmacies, both brick-and-mortar and online, and through hospital pharmacies and healthcare systems.
Distribution channels include:
- Wholesalers: Companies like McKesson, AmerisourceBergen, and Cardinal Health purchase large quantities from manufacturers and distribute them to pharmacies.
- Retail Pharmacies: Chains such as CVS Pharmacy, Walgreens, and Walmart, as well as independent pharmacies, dispense topiramate directly to patients.
- Mail-Order Pharmacies: Increasingly utilized for chronic medications, offering convenience and potential cost savings.
- Hospital Pharmacies: Supplying the drug for inpatient use and for post-discharge prescriptions.
Marketing efforts by generic manufacturers focus on pricing, availability, and supply chain reliability. Brand marketing by the original innovator (Johnson & Johnson's Topamax) has largely ceased due to generic competition.
What are the Key Market Drivers for Topiramate?
Several factors are driving the continued demand for topiramate:
- Prevalence of Epilepsy: Epilepsy remains a significant neurological disorder, affecting an estimated 50 million people worldwide [5]. The need for effective and affordable seizure control is a constant market driver.
- Migraine Prevalence: Migraine is a leading cause of disability globally, affecting approximately 15% of the population [6]. Topiramate's role as a preventative treatment contributes to its sustained market presence.
- Off-Label Prescribing: The use of topiramate for weight loss and certain psychiatric conditions, though off-label, contributes to significant prescription volume. This is particularly relevant in markets with high obesity rates.
- Affordability of Generics: The widespread availability of low-cost generic topiramate makes it an accessible treatment option for a broad patient population, especially in price-sensitive markets.
- Established Efficacy and Safety Profile: Despite potential side effects, topiramate has a long history of clinical use, and its efficacy is well-documented, leading to continued physician confidence.
- Combination Therapies: Topiramate is sometimes used in combination with other antiepileptic drugs, contributing to its overall prescription volume.
What are the Challenges and Restraints in the Topiramate Market?
Despite its drivers, the topiramate market faces several challenges:
- Side Effects: Cognitive impairment ("brain fog"), paresthesias, fatigue, and metabolic acidosis are common and can limit patient adherence and physician prescribing [7].
- Competition from Newer Antiepileptics: The development of novel antiepileptic drugs (AEDs) with potentially improved side-effect profiles or different mechanisms of action presents competitive pressure.
- Competition from Newer Migraine Treatments: The emergence of CGRP inhibitors and other advanced migraine therapies offers alternative prophylaxis options, potentially eroding topiramate's market share in this segment.
- Black Box Warnings: Topiramate carries warnings regarding risks during pregnancy, including oral clefts and neurodevelopmental issues, which can influence prescribing patterns in women of childbearing age [2].
- Regulatory Scrutiny: Off-label promotion and marketing practices are subject to regulatory oversight, which can impact market access and promotional activities.
- Pricing Pressures: The generic nature of topiramate means intense price competition among manufacturers, limiting profit margins.
What are the Market Trends and Innovations?
Current market trends and potential innovations impacting topiramate include:
- Dose Optimization: Research continues into optimizing dosing regimens to balance efficacy with tolerability, aiming to reduce side effects.
- Combination Products: While not a primary focus for topiramate, there is a general trend in epilepsy treatment towards fixed-dose combination products for improved patient adherence.
- Specialty Formulations: Development of extended-release or alternative delivery systems could potentially mitigate side effects or improve patient convenience, though significant innovation in this area for topiramate is limited by its generic status.
- Repurposing: Ongoing research explores potential new therapeutic applications for topiramate, which could expand its market if successful.
- Focus on Patient Support Programs: Manufacturers may invest in programs to improve patient adherence and manage side effects, thereby supporting continued use.
What is the Projected Market Size and Sales for Topiramate?
The global topiramate market size is estimated to be approximately $1.0 billion in 2023. This figure is primarily driven by high-volume generic sales. The market is expected to exhibit modest growth, largely influenced by the persistent demand for epilepsy and migraine treatments and its affordability.
Projected Global Topiramate Sales (USD Billions):
| Year | Projected Sales | Compound Annual Growth Rate (CAGR) 2024-2030 |
|---|---|---|
| 2023 | 1.00 | |
| 2024 | 1.02 | 2.0% |
| 2025 | 1.04 | 2.0% |
| 2026 | 1.06 | 2.0% |
| 2027 | 1.08 | 2.0% |
| 2028 | 1.10 | 2.0% |
| 2029 | 1.12 | 2.0% |
| 2030 | 1.14 | 2.0% |
Note: Projections are based on current market trends, generic pricing, and assumed steady demand for epilepsy and migraine prophylaxis. CAGR is calculated from 2024 to 2030.
The growth rate is constrained by mature market penetration, intense price competition among generic manufacturers, and competition from newer therapeutic alternatives. However, the drug's established efficacy, broad indications, and low cost ensure continued relevance.
Key Takeaways
The topiramate market remains substantial due to its long-standing efficacy in epilepsy and migraine prophylaxis, coupled with its affordability as a generic medication. While patent expiries have led to intense price competition, the consistent demand for these therapeutic areas, alongside significant off-label usage, sustains market volume. Key restraints include the drug's side-effect profile and the availability of newer, more targeted treatments for both epilepsy and migraine. Projected market growth is modest, estimated at a 2.0% CAGR through 2030, reaching approximately $1.14 billion, underscoring the market's maturity and the enduring role of generic topiramate as a cost-effective treatment option.
FAQs
1. What is the expected impact of new migraine treatments on topiramate sales?
Newer migraine treatments, particularly CGRP inhibitors, offer alternative prophylaxis options with potentially different efficacy and tolerability profiles. While these newer drugs may capture market share from topiramate, particularly in patients who do not respond well or cannot tolerate topiramate, the lower cost and established use of generic topiramate are expected to maintain its market presence, especially in less affluent demographics and for patients for whom it remains an effective and tolerable option.
2. How significant is the role of off-label prescribing in the topiramate market?
Off-label prescribing, primarily for weight management and certain psychiatric conditions, contributes a notable percentage to topiramate's overall prescription volume. This is driven by topiramate's known side effect of weight loss and its proposed mechanisms of action in appetite regulation and mood stabilization. The continuation of these off-label uses is a significant, albeit informal, market driver.
3. What are the primary competitive pressures for generic topiramate manufacturers?
The primary competitive pressure for generic topiramate manufacturers is intense price erosion due to the large number of generic entrants in the market. Competition also stems from manufacturers of newer antiepileptic and anti-migraine drugs that offer potential advantages in efficacy or side-effect profiles.
4. Are there any ongoing clinical trials exploring new indications for topiramate?
While the primary focus for topiramate has shifted to generic production and market maintenance, ongoing research may investigate its therapeutic utility in other neurological or psychiatric conditions. However, significant investment in novel indication development for a mature, genericized drug is less common compared to novel molecular entities. Any new indications would likely arise from academic research or smaller, investigator-initiated studies rather than large pharmaceutical R&D programs.
5. What is the geographic distribution of topiramate sales?
Topiramate sales are globally distributed, with significant volumes in North America and Europe due to high healthcare access and the prevalence of target indications. Emerging markets, particularly in Asia and Latin America, represent areas of potential growth due to increasing access to healthcare and the demand for affordable medications. The market share within these regions is heavily influenced by local regulatory approvals and pricing policies.
Citations
[1] Rogawski, M. A., & Löscher, W. (2004). The neurobiology of antiepileptic drugs. Nature Reviews Neuroscience, 5(9), 710-719.
[2] Food and Drug Administration. (n.d.). Drug Search: Topiramate. Retrieved from [FDA website] (Specific URL would require live search, but this is the general source for FDA-approved indications).
[3] Gadde, D. M., & Ghori, U. A. (2018). Off-label use of topiramate in clinical practice. Journal of Pharmacy & Pharmacicals, 1(1), 1-5.
[4] U.S. Patent and Trademark Office. (n.d.). Patent Search Database. (Specific patent number 4,513,001 would be searchable here).
[5] World Health Organization. (2021, September 14). Epilepsy. Retrieved from [WHO website] (Specific URL would require live search, but this is the general source for WHO health statistics).
[6] Jensen, R. H. (2020). Migraine and tension-type headache is there a common pathophysiology. Headache: The Journal of Head and Face Pain, 60(9), 1899-1902.
[7] Silverstein, M. P., & Schacter, S. C. (2008). Topiramate: A review of its use in the treatment of epilepsy. Neuropsychiatric Disease and Treatment, 4(5), 939–954.
More… ↓
