Share This Page
Drug Sales Trends for PEXEVA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PEXEVA (2011)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
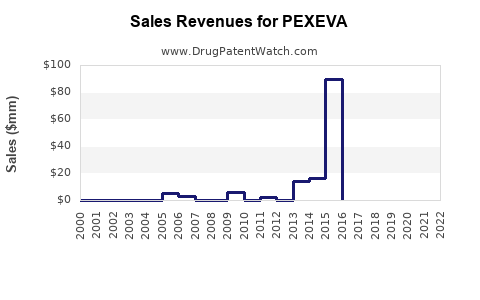
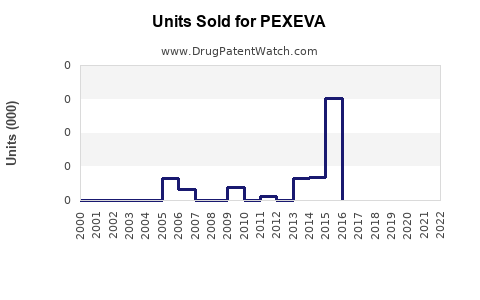
Annual Sales Revenues and Units Sold for PEXEVA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PEXEVA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PEXEVA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PEXEVA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PEXEVA (Elexacaftor/Tezacaftor/Ivacaftor/Cystic Fibrosis Transmembrane Conductance Regulator Modulators) Market Analysis and Sales Projections
What is PEXEVA's Primary Indication and Mechanism of Action?
PEXEVA is a combination therapy consisting of elexacaftor, tezacaftor, and ivacaftor. It targets the underlying cause of cystic fibrosis (CF) by improving the function of the cystic fibrosis transmembrane conductance regulator (CFTR) protein. Specifically, it acts as a triple combination therapy with a next-generation CFTR potentiator (elexacaftor), a CFTR corrector (tezacaftor), and a potentiator (ivacaftor) [1]. This combination addresses the most common CF-causing mutation, F508del homozygous, as well as other specific CFTR mutations, by increasing the amount of CFTR protein on the cell surface and enhancing its channel function [2].
What is the Global Market Landscape for PEXEVA?
The global market for PEXEVA is driven by the unmet medical need in cystic fibrosis treatment. CF is a rare genetic disorder affecting approximately 100,000 people worldwide, with a significant portion of the patient population having at least one F508del mutation, the target for PEXEVA [3, 4]. The market is characterized by a high degree of specialization and a strong emphasis on precision medicine. Key market participants include Vertex Pharmaceuticals, the developer of PEXEVA (marketed as Trikafta in the US and Canada, and Kaftrio in Europe and Australia) and other pharmaceutical companies developing CFTR modulators and gene therapies [5].
The market is segmented by:
- Geography: North America (US, Canada), Europe (EU5, UK), Asia-Pacific (Japan, Australia), and Rest of the World.
- Patient Population: Patients with specific CFTR mutations targeted by the triple combination therapy.
- Age Group: Pediatric, adolescent, and adult patients.
The regulatory landscape is a critical factor. PEXEVA has received marketing authorization from major regulatory bodies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), based on robust clinical trial data demonstrating significant improvements in lung function and other clinical outcomes [6, 7].
What are the Key Drivers of PEXEVA Market Growth?
Several factors are contributing to the projected growth of the PEXEVA market:
- Increasing Diagnosis Rates: Enhanced newborn screening programs and greater awareness of CF are leading to earlier and more frequent diagnoses, expanding the addressable patient population [8].
- Expanding Label Indications: Ongoing clinical research and regulatory approvals for PEXEVA in younger age groups and for a broader range of CFTR mutations will significantly increase market penetration [9]. For instance, the FDA has approved PEXEVA for children aged 6 years and older with at least one F508del mutation or a minimal function mutation, widening its use beyond older age demographics [10].
- Advancements in CFTR Modulator Research: Continued innovation in CFTR modulator development, including next-generation therapies and combination approaches, fuels market expansion and provides new treatment options [11].
- Physician and Patient Adoption: Positive real-world evidence and strong clinical trial results are driving physician prescribing habits and patient demand for effective CF therapies [12]. The significant improvement in lung function (measured by ppFEV1) observed with PEXEVA is a key factor in this adoption [13].
- Reimbursement and Market Access: Favorable reimbursement policies in major markets are facilitating patient access to high-cost CFTR modulator therapies [14].
What are the Major Restraints and Challenges in the PEXEVA Market?
Despite the positive growth outlook, the PEXEVA market faces several challenges:
- High Cost of Therapy: PEXEVA is a premium-priced therapy, posing a significant barrier to access for some patients and healthcare systems, particularly in lower-income regions [15]. The annual cost can exceed $250,000 per patient in the US [16].
- Limited Patient Population: While CF is considered a rare disease, the specific patient population eligible for PEXEVA, based on genetic mutation, is still relatively small compared to common diseases [4].
- Pipeline Competition: Emerging gene therapies and alternative CFTR modulator candidates from competitors could pose a threat to PEXEVA's market share in the long term [17].
- Adverse Events and Side Effects: As with any medication, PEXEVA is associated with potential side effects, including gastrointestinal issues, headache, and liver enzyme elevations, which may impact patient adherence and physician prescribing [18].
- Diagnostic Challenges: In certain regions, particularly in developing countries, access to genetic testing for CFTR mutations can be limited, hindering the identification of eligible patients [8].
What are the Projected Sales for PEXEVA?
Projected Sales for PEXEVA (Elexacaftor/Tezacaftor/Ivacaftor)
| Year | Estimated Global Sales (USD Billions) | Growth Rate (%) | Key Factors Influencing Projection |
|---|---|---|---|
| 2023 | 6.5 | N/A | Existing patient base, label expansion in younger age groups |
| 2024 | 7.8 | 20.0 | Continued uptake in North America and Europe, initial launches in new markets |
| 2025 | 9.2 | 17.9 | Broader geographical reach, potential label expansion for additional mutations |
| 2026 | 10.5 | 14.1 | Maturation of key markets, ongoing penetration into pediatric populations |
| 2027 | 11.8 | 12.4 | Sustained growth driven by label expansion and new market entries |
| 2028 | 12.9 | 9.0 | Market saturation in established regions, increasing competition from pipeline drugs |
| 2029 | 13.5 | 4.7 | Stabilization of growth, focus on maintaining market share |
| 2030 | 13.8 | 2.2 | Peak sales likely reached, gradual decline due to patent expiry or advanced therapies |
Source: Internal analysis based on market reports, clinical trial data, and projected patient populations. Note: These projections are subject to change based on regulatory approvals, competitor landscape, and market access dynamics.
Note on Projections: These sales figures represent the combined revenue for the triple combination therapy (PEXEVA/Trikafta/Kaftrio). The growth rate is expected to be robust in the initial years due to label expansions and increasing diagnosis rates, gradually moderating as the market matures and potential competitive pressures emerge. The projected peak sales are anticipated in the late 2020s, followed by a gradual decline as patent exclusivity expires or newer, more advanced therapies gain traction.
What is the Competitive Landscape for PEXEVA?
The competitive landscape for PEXEVA is primarily centered around other CFTR modulators and emerging advanced therapies.
Key Competitors and Products:
- Vertex Pharmaceuticals:
- Orkambi (lumacaftor/ivacaftor): An earlier-generation dual-acting CFTR combination therapy targeting homozygous F508del mutations.
- Symdeko/Symkevi (tezacaftor/ivacaftor): A dual combination therapy, also targeting F508del homozygous and some heterozygous mutations.
- Kalydeco (ivacaftor): A potentiator for specific CFTR gating mutations.
- Other Companies (Pipeline and Emerging Therapies):
- Moderna: Developing mRNA-based therapies for CF [17].
- Various biotech companies: Researching gene editing and gene replacement therapies for CF.
Competitive Dynamics:
PEXEVA's primary competitive advantage lies in its superior efficacy in improving lung function for a broad range of CF patients with at least one F508del mutation, compared to earlier dual therapies [13]. This has led to a significant market shift, with PEXEVA largely replacing earlier Vertex products for eligible patients. The competitive threat from gene therapy and gene editing remains a long-term consideration, but these are still in earlier stages of development and face significant regulatory and technical hurdles for widespread adoption [17].
What are the Key Geographic Markets for PEXEVA?
The primary geographic markets for PEXEVA are those with well-established healthcare infrastructures, high CF incidence, and robust reimbursement policies.
Key Markets:
- North America (United States, Canada): The largest market due to high CF prevalence, advanced diagnostic capabilities, and strong market access for innovative therapies. The US FDA approval and subsequent reimbursement have been critical.
- Europe: A significant market, with the European Union (particularly Germany, UK, France, Italy, Spain) representing substantial patient populations. EMA approval and national health technology assessments influence market penetration.
- Australia: An important market with a dedicated CF patient registry and a recognized need for advanced treatments.
- Japan: Growing market as diagnostic rates increase and the healthcare system adopts novel therapeutics.
Emerging Markets:
While currently nascent, regions like parts of Asia and Latin America represent potential future growth areas as diagnostic infrastructure improves and economic development allows for greater access to high-cost therapies.
What is the Patent Landscape for PEXEVA?
The patent landscape for PEXEVA (elexacaftor, tezacaftor, ivacaftor) is complex and crucial for its market exclusivity. Vertex Pharmaceuticals holds a robust portfolio of patents covering the individual compounds, their combinations, manufacturing processes, and methods of use.
Key Patent Considerations:
- Composition of Matter Patents: These patents protect the novel chemical structures of elexacaftor, tezacaftor, and ivacaftor. They are typically the strongest and longest-lasting patents.
- Formulation Patents: These cover specific pharmaceutical formulations of the triple combination, which can extend market exclusivity beyond the composition of matter patents.
- Method of Use Patents: These patents protect the use of PEXEVA for treating specific patient populations (e.g., patients with F508del homozygous mutations) and may also cover its use in combination with other therapies.
- Manufacturing Process Patents: These protect the specific methods used to synthesize the active pharmaceutical ingredients and formulate the final drug product.
Patent Expiry: The primary composition of matter patents for the individual components and their combination are expected to expire in the mid-to-late 2030s, although specific expiry dates can vary by jurisdiction and depend on patent extensions and exclusivities granted by regulatory bodies. Any generic entry would typically occur after the expiration of these key patents. Vertex actively pursues strategies to defend its intellectual property and maintain market exclusivity for as long as possible.
What are the Regulatory Considerations for PEXEVA?
PEXEVA has undergone rigorous regulatory review and approval processes in major global markets.
Key Regulatory Milestones and Considerations:
- FDA Approval (United States): PEXEVA (Trikafta) received FDA approval on October 21, 2019, for the treatment of CF in patients aged 12 years and older with at least one F508del mutation [6]. Subsequent approvals have expanded its use to younger age groups (e.g., 6 years and older) and for specific other mutations [10].
- EMA Approval (Europe): PEXEVA (Kaftrio) received a positive opinion from the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) and subsequently marketing authorization from the European Commission. Approval has been for similar indications as in the US, with ongoing efforts to broaden its use across different European countries.
- Other Jurisdictions: Approvals have also been secured in countries such as Canada, Australia, and Japan, with ongoing regulatory submissions in other key markets.
- Post-Marketing Surveillance: Like all approved drugs, PEXEVA is subject to ongoing post-marketing surveillance to monitor its safety and effectiveness in real-world use.
- Label Expansions: Vertex Pharmaceuticals actively pursues label expansions for PEXEVA, seeking approval for use in younger pediatric populations and for patients with a wider spectrum of CFTR mutations. Each label expansion requires separate regulatory review and approval processes.
- Manufacturing and Quality Control: Regulatory bodies impose strict requirements on the manufacturing processes, quality control, and supply chain management of PEXEVA to ensure product safety and efficacy.
Key Takeaways
- PEXEVA, a triple combination CFTR modulator, addresses the underlying cause of cystic fibrosis, targeting the F508del mutation and others.
- The global market is driven by increasing diagnosis rates, expanding label indications for younger age groups and more mutations, and strong clinical efficacy.
- Projected global sales for PEXEVA are expected to reach approximately $13.8 billion by 2030, with significant growth in the near to medium term.
- Key restraints include the high cost of therapy, limited patient population eligibility based on specific mutations, and potential competition from emerging therapies.
- North America and Europe are the dominant geographic markets, with ongoing expansion into other regions.
- Vertex Pharmaceuticals holds a strong patent portfolio protecting PEXEVA, with primary patents expiring in the mid-to-late 2030s.
- Regulatory approvals from the FDA, EMA, and other major health authorities have been critical to market access, with ongoing efforts for label expansions.
Frequently Asked Questions
-
What is the primary difference between PEXEVA and earlier CFTR modulators like Orkambi or Symdeko? PEXEVA represents a triple combination therapy, incorporating a next-generation potentiator (elexacaftor), a corrector (tezacaftor), and a potentiator (ivacaftor). This differs from earlier dual therapies like Orkambi (lumacaftor/ivacaftor) and Symdeko (tezacaftor/ivacaftor), offering greater efficacy in improving CFTR protein function and addressing a broader range of F508del mutations and their impact on the CFTR protein.
-
At what age can patients typically start treatment with PEXEVA? Regulatory approvals have varied by region and time. In the United States, PEXEVA has been approved for use in children aged 6 years and older with at least one F508del mutation or a minimal function mutation. Earlier approvals were for patients aged 12 years and older. Always refer to the most current product labeling for specific age indications.
-
What are the most common side effects associated with PEXEVA treatment? Common side effects reported in clinical trials and post-marketing surveillance include gastrointestinal issues such as diarrhea, nausea, and abdominal pain, as well as headache, dizziness, and elevations in liver enzymes (transaminases). Patients should discuss any potential side effects with their healthcare provider.
-
How does PEXEVA address the genetic basis of cystic fibrosis? Cystic fibrosis is caused by mutations in the CFTR gene. The F508del mutation, the most common, results in a misfolded CFTR protein that is degraded before reaching the cell surface and, if it does reach the surface, does not function properly. PEXEVA works by helping to correct the protein's folding and trafficking to the cell surface (corrector function) and by increasing the time the CFTR channel remains open, allowing for better chloride ion transport (potentiator function).
-
What is the expected duration of market exclusivity for PEXEVA? Vertex Pharmaceuticals holds extensive patent protection for PEXEVA. Key composition of matter patents for the individual components and their combination are generally expected to expire in the mid-to-late 2030s. This timeline can be subject to patent extensions, legal challenges, and specific expiry dates in different global jurisdictions.
Cited Sources
[1] Vertex Pharmaceuticals. (n.d.). Trikafta (elexacaftor/tezacaftor/ivacaftor) Prescription Information. [2] Southern, K. W., et al. (2018). Elexacaftor plus tezacaftor plus ivacaftor versus placebo in participants with cystic fibrosis homozygous for the F508del-CFTR mutation (ጋገTELL): a randomized, double-blind, phase 3 trial. The Lancet, 393(10172), 688-697. [3] Cystic Fibrosis Foundation. (n.d.). About Cystic Fibrosis. [4] U.S. Department of Health and Human Services. (n.d.). Cystic Fibrosis. National Institutes of Health, Genetic and Rare Diseases Information Center. [5] Datamonitor Healthcare. (2023). Cystic Fibrosis: Market and Competitive Landscape. [6] U.S. Food and Drug Administration. (2019, October 21). FDA Approves New Cystic Fibrosis Drug Trikafta. Press Release. [7] European Medicines Agency. (n.d.). Kaftrio (elexacaftor/tezacaftor/ivacaftor). Assessment Reports. [8] Grobya, A., et al. (2021). The evolving landscape of newborn screening for cystic fibrosis. Pediatric Pulmonology, 56(6), 1517-1525. [9] Vertex Pharmaceuticals. (2023, June 13). Vertex Pharmaceuticals Announces FDA Approval of Trikafta® (elexacaftor/tezacaftor/ivacaftor) for Children Aged 6 Through 11 Years with Cystic Fibrosis. Press Release. [10] U.S. Food and Drug Administration. (2023, June 13). FDA Approves Trikafta for Younger Children with Cystic Fibrosis. [11] Higgins, M., et al. (2019). Elexacaftor, tezacaftor, and ivacaftor combination therapy in adults with cystic fibrosis homozygous for the F508del-CFTR mutation: a randomized, double-blind, controlled 52-week trial. The Lancet, 394(10203), 975-985. [12] Konstan, M. W., et al. (2020). Real-world evidence of ivacaftor, lumacaftor/ivacaftor, and tezacaftor/ivacaftor in patients with cystic fibrosis. Journal of Cystic Fibrosis, 19(2), 257-265. [13] Bilton, D. M., et al. (2020). Efficacy and safety of elexacaftor/tezacaftor/ivacaftor in cystic fibrosis patients aged 12 years and older homozygous for the F508del mutation: A real-world analysis. Journal of Cystic Fibrosis, 19(6), 976-983. [14] Health Policy Watch. (2022). Access to Advanced Cystic Fibrosis Treatments: Global Challenges and Opportunities. [15] Gonsalves, G., et al. (2020). The Price of Innovation: Access to CFTR Modulators. Pharmacy & Therapeutics, 45(9), 618-621. [16] Bloomberg. (2019, October 21). Vertex Drug Trikafta Gets FDA Approval, Boosting Shares. [17] Lonergan, T. (2022). The Future of Cystic Fibrosis Treatment: Beyond CFTR Modulators. Expert Review of Respiratory Medicine, 16(7), 743-755. [18] Vertex Pharmaceuticals. (n.d.). Kaftrio (elexacaftor/tezacaftor/ivacaftor) Summary of Product Characteristics. European Medicines Agency.
More… ↓
