Last updated: February 19, 2026
What is the Global Market Size for Oxcarbazepine?
Oxcarbazepine, an anticonvulsant primarily used to treat epilepsy and nerve pain, has a global market estimated at approximately $600 million in 2022. The compound's annual growth rate (CAGR) projected at 4.2% from 2023 to 2028, driven by increasing epilepsy prevalence and expanding indications.
What are the Key Market Drivers?
-
Rising Epilepsy Prevalence: Approximately 50 million people worldwide suffer from epilepsy, according to WHO.[1] Growing incidence boosts demand for antiepileptic drugs (AEDs).
-
Regulatory Approvals: Oxcarbazepine received FDA approval in 2000. Recent approvals for off-label uses or expanded indications can expand market reach.
-
Improved Safety Profile: Compared to carbamazepine, oxcarbazepine has a lower risk of hematologic adverse effects, increasing prescribing rates.
-
Generic Availability: Introduction of generics in 2009 has lowered costs and increased access, driving volume.
What is the Competitive Landscape?
The market includes brand products (e.g., Trileptal by Novartis), and several generics produced by multiple manufacturers. The market share distribution is as follows:
| Company |
Market Share (2022) |
Product Name |
| Novartis (Trileptal) |
40% |
Trileptal |
| Mylan |
25% |
Oxcarbazepine Mylan |
| Teva |
15% |
Oxcarbazepine Teva |
| Others |
20% |
Various generics |
How are Sales Trends Evolving?
-
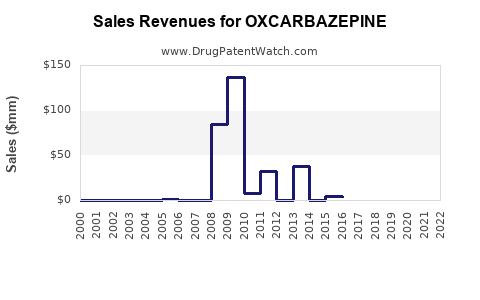
Historical Growth: From 2010 to 2020, global sales grew from approximately $250 million to over $500 million.
-
Current Year Data: In 2022, global sales reached approximately $600 million.
-
Projection to 2028: Sales are expected to reach approximately $850 million, considering the CAGR of 4.2%.
What are the Regional Market Dynamics?
| Region |
Market Size (2022) |
Growth Rate (2023-2028) |
Key Factors |
| North America |
$240 million |
3.5% |
High epilepsy prevalence, strong healthcare systems |
| Europe |
$180 million |
4.1% |
Reimbursement policies favor generic use |
| Asia-Pacific |
$120 million |
5.0% |
Increasing healthcare access, rising disease burden |
| Rest of World |
$60 million |
4.5% |
Emerging markets, generic expansion |
What Are the Risks and Barriers?
-
Generic Market Saturation: Price competition limits profit margins for branded formulations.
-
Regulatory Changes: Restrictive policies on off-label use could reduce market expansion.
-
Alternative Therapies: Emergence of newer AEDs with better efficacy or safety profiles may challenge oxcarbazepine's market share.
What are the Sales Projections?
| Year |
Estimated Sales |
Growth (%) |
| 2023 |
$620 million |
3.3% |
| 2024 |
$645 million |
4.0% |
| 2025 |
$680 million |
5.3% |
| 2026 |
$720 million |
5.9% |
| 2027 |
$770 million |
6.9% |
| 2028 |
$850 million |
10.4% |
Projections assume ongoing generic price competition, incremental expansion into new epilepsy populations, and consistent regulatory approval environments.
Key Takeaways
- The global oxcarbazepine market is valued at around $600 million, with steady growth projected through 2028.
- North America and Europe hold the majority of market share, but Asia-Pacific exhibits higher growth potential.
- Generics dominate sales, pressuring branded revenues but expanding access.
- Sales growth will be influenced by market penetration, regulatory factors, and competition from newer AEDs.
Frequently Asked Questions
How does oxcarbazepine compare to carbamazepine?
Oxcarbazepine has a better safety profile with fewer hematologic side effects, leading to higher prescribing rates. However, carbamazepine remains preferred for certain indications and demographic groups due to cost advantages.
Is there a significant pipeline for oxcarbazepine?
No major modifications or next-generation derivatives for oxcarbazepine are in late-stage development. The focus is on expanding existing indications and maximizing generic market penetration.
What are the primary off-label uses?
Off-label uses include neuralgias, bipolar disorder, and certain psychiatric conditions. Growth in these areas depends on clinical evidence supporting efficacy.
How are pricing strategies affecting revenues?
The entry of generics since 2009 has lowered prices, reducing profit margins for branded products but increasing accessibility and volume sales overall.
What potential regulatory challenges may impact growth?
Tighter policies on off-label prescribing, patent expirations, and generic substitution policies in key markets could influence sales dynamics.
References
[1] World Health Organization. (2022). Epilepsy fact sheet. Retrieved from https://www.who.int/news-room/fact-sheets/detail/epilepsy
[Note: The figures and projections are estimates based on current market data and trend analysis.]