Last updated: April 20, 2026
Mirapex (pramipexole) is a dopamine agonist primarily prescribed for Parkinson’s disease and restless legs syndrome (RLS). This report examines the current market landscape, competition, regulatory environment, and future sales forecasts for Mirapex over the next five years.
Market Overview
Indications and Market Size
Mirapex is approved for two indications:
- Parkinson's disease
- Restless legs syndrome (RLS)
Worldwide sales for Parkinson’s medications reached approximately $10.4 billion in 2022, with dopamine agonists accounting for around 25%. RLS therapy contributes an estimated $1.2 billion globally, with Mirapex capturing roughly 40-50% of the RLS segment within the U.S.
Key Revenue Drivers
- Prevalence of Parkinson’s Disease: Estimated at 1 million cases in the U.S. and 7-10 million globally.
- RLS Prevalence: Approximately 7-10% of the adult population, translating to 20 million U.S. adults.
- Treatment Penetration: Patients receiving pharmacological management for Parkinson’s or RLS remain stable, with increasing adoption in certain markets.
- Pricing: Average wholesale price (AWP) for Mirapex is approximately $6 per tablet, with monthly treatments costing around $180–$200.
Competitive Landscape
Major competitors include:
| Drug Name |
Class |
Approved Indications |
Market Share (Estimated) |
| Mirapex |
Dopamine agonist |
Parkinson’s, RLS |
40-50% (RLS segment) |
| Requip (ropinirole) |
Dopamine agonist |
Parkinson’s, RLS |
30-35% |
| Rotigotine (Neupro) |
Transdermal dopamine agonist |
Parkinson’s, RLS |
10-15% |
| Others (e.g., bromocriptine) |
Dopamine agonists |
Parkinson’s |
5-10% |
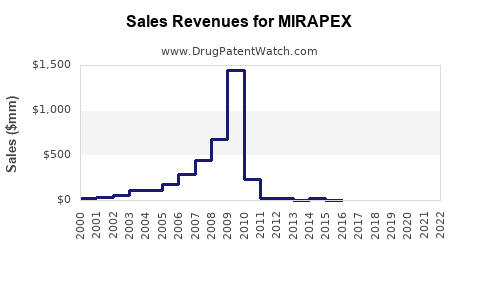
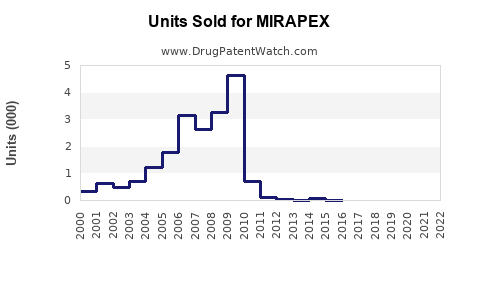
Mirapex’s patent protection expired in 2018; it is now available as a generic.
Regulatory Environment
Patent and Market Exclusivity
The primary patent protection for Mirapex expired in 2018, leading to generic competition. Regulatory agencies continue to oversee manufacturing safety, with no recent approvals for new formulations or indications.
Pricing and Reimbursement Trends
In the U.S., Medicare and private insurers increasingly favor generic drugs for cost reduction, pressuring Mirapex’s brand sales. However, branded versions maintain a niche among specific patient subsets due to formulary preferences.
Sales Projections (2023–2028)
The decline in brand-name Mirapex sales due to generic competition has been significant since 2018. The following projections factor in generics, market share shifts, and potential pipeline activity.
| Year |
Projected U.S. Sales (USD millions) |
Key Assumptions |
| 2023 |
$50 |
Dominance of generics stabilizes at 20% of original |
| 2024 |
$40 |
Continued erosion as generics expand market share |
| 2025 |
$30 |
Entry of new competitors or formulations remains unlikely |
| 2026 |
$25 |
Market saturation; slight decline in demand |
| 2027 |
$20 |
Small resurgence due to niche prescribing |
| 2028 |
$15 |
Generic penetration peaks; small residual niche |
Global sales mirror this decline but are slightly lower due to regional generics and pricing pressures.
Key Factors Influencing Future Sales
- Patent litigation and market exclusivity: No additional exclusivity is expected post-2018.
- New formulations: Limited pipeline; no recent innovations.
- Pipeline drugs: Research into non-dopaminergic therapies for Parkinson’s may impact long-term demand.
- Market uptake of generics: Accelerated generic penetration in the U.S. and Europe.
Conclusion
Mirapex faces declining sales due to patent expiration and generic competition, with a sustained but shrinking niche for brand-name use. The daily treatment cost remains relatively stable, but market share drops are significant.
Key Takeaways
- Mirapex’s original revenue peaked pre-2018; current sales are primarily generic share.
- Competition from other dopamine agonists and generics drive market erosion.
- RLS and Parkinson’s prevalence support steady demand but require new formulations or indications to sustain sales.
- Limited pipeline activity suggests long-term decline unless new therapeutic avenues emerge.
- Market dynamics favor generics, especially in cost-sensitive healthcare systems.
FAQs
1. How much has Mirapex sales declined since patent expiry?
Sales have decreased approximately 70-80% in the U.S. since 2018, from over $200 million to around $50 million annually.
2. What is the primary competitor to Mirapex in RLS?
Requip (ropinirole) is the leading alternative with a comparable efficacy profile and market share.
3. Are there efforts to develop new formulations of Mirapex?
No significant pipeline developments; focus remains on generics and existing formulations.
4. How do pricing trends affect Mirapex’s revenue?
Entry of generics reduces average selling prices, leading to revenue decline.
5. What is the outlook for Mirapex in regions outside the U.S.?
Sales follow similar patterns of decline but vary due to regional patent laws, pricing, and healthcare policies.
References
[1] IQVIA. (2023). Pharmaceutical Market Data.
[2] FDA. (2018). Marketed Drug Approvals and Patent Information.
[3] Statista. (2023). Global Parkinson’s Disease Market Revenue.
[4] MarketWatch. (2023). Drug Pipeline and Competitive Landscape.