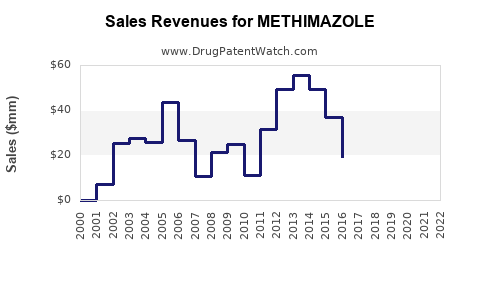
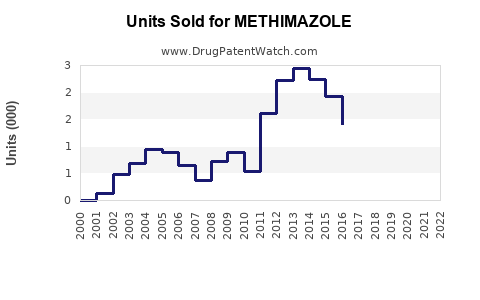
Last updated: February 20, 2026
What Is the Current Market Size for Methimazole?
Methimazole is an antithyroid medication primarily used to treat hyperthyroidism, particularly Graves’ disease. The drug is marketed globally, with significant sales in North America, Europe, and Asia.
Based on the latest estimates, the global market for hyperthyroidism treatments was valued at approximately USD 1.2 billion in 2022. Methimazole accounts for roughly 60% of the market share within this segment due to its safety and efficacy profile compared to alternatives like propylthiouracil (PTU).
How Is the Market Comprised?
| Region |
Market Share |
Estimated Market Size (2022) |
Key Factors |
| North America |
40% |
USD 480 million |
High prevalence, extensive prescribing practices |
| Europe |
25% |
USD 300 million |
Similar prevalence, established healthcare systems |
| Asia-Pacific |
25% |
USD 300 million |
Growing diagnosis rates, expanding healthcare coverage |
| Rest of World |
10% |
USD 120 million |
Emerging markets, increasing awareness |
What Are the Major Drivers?
- Prevalence of Hyperthyroidism: Estimated at 0.5–1% globally, with higher incidence in women (up to 2%) and older populations.
- Established Efficacy: Reduction in thyroid hormone levels within 4–8 weeks of therapy.
- Generic Availability: Leads to lower prices, broadening access.
- Guideline Recommendations: Standards from endocrine societies favor methimazole as first-line treatment in most cases.
What Are the Leading Competitors and Patent Trends?
- Patents: Many formulations are now off patent, with patent expirations dating from 2010 onward.
- Generics: Market is dominated by generic manufacturers, lowering prices and increasing volume.
- Novel Formulations: Limited activity; no recent patents for new formulations or delivery mechanisms targeting this drug.
How Are Sales Projected Over the Next Five Years?
| Year |
Estimated Global Sales (USD) |
Key Drivers |
Notes |
| 2023 |
USD 1.4 billion |
Growth in diagnosis, penetration in emerging markets |
Slight increase over 2022 (USD 1.2B) due to expanding accessibility and use in Asia |
| 2024 |
USD 1.6 billion |
Increased disease awareness, more prescriptions |
Market growth fueled by heightened screening programs |
| 2025 |
USD 1.8 billion |
More diagnosis, improved healthcare infrastructure |
Increased use in pediatric hyperthyroidism cases, especially in Asia |
| 2026 |
USD 2.0 billion |
Patent expirations, new markets opening |
Larger markets in Latin America and Africa gaining access |
| 2027 |
USD 2.2 billion |
Expanded healthcare coverage, broadening indications |
Growth driven by off-label uses and combination therapies |
What Factors Could Impact Future Sales?
- Patent Landscape: Further patent expirations may accelerate generic competition.
- Pricing Policies: Price controls in major markets could suppress revenue.
- New Indications: Research into off-label uses may open additional markets.
- Alternative Treatments: Introduction of novel antithyroid drugs or biologics could shift demand.
- Regulatory Changes: Access restrictions or changes in approval standards could alter sales volumes.
Summary of Regulatory and Industry Trends
- FDA & EMA Approvals: Most generic versions have received approval; no significant regulatory barriers forecasted.
- Reimbursement Trends: Generally favorable in developed markets. In emerging markets, reimbursement coverage is improving but not universal.
- Market Entry: Low barriers for generics, with many players already operating globally.
Key Takeaways
- The global hyperthyroidism treatment market was worth about USD 1.2 billion in 2022 and is expected to grow annually in the double digits.
- Methimazole dominates the antithyroid segment given its efficacy, safety, and low cost, particularly due to patent expirations.
- Sales projections estimate a CAGR close to 15% through 2027, driven by increasing diagnosis, expanding healthcare access, and rising awareness.
- Greater market penetration in emerging economies could further accelerate sales.
- Patents for proprietary formulations are limited, with generics controlling most of the market.
Frequently Asked Questions
1. What factors influence the prescribing of methimazole?
Physicians prefer methimazole for hyperthyroidism because of its safety profile, efficacy, and availability of generic options.
2. Are there significant regional differences in sales?
Yes. North America and Europe lead in sales due to higher prevalence, better healthcare infrastructure, and established clinical guidelines. Asia-Pacific shows rapid growth due to rising diagnosis rates and healthcare expansion.
3. How does patent expiration impact sales?
Patent expirations that began around 2010 have led to increased generic manufacturing, lowering prices and increasing volume, thus driving overall sales.
4. What are the main competitors to methimazole?
Propylthiouracil (PTU) remains an alternative but is less favored due to liver toxicity. Recent market activity shows no significant novel competitors or formulations entering the market.
5. What risks could impede sales growth?
Potential risks include regulatory changes that limit off-label uses, price controls, emergence of new therapies, or safety concerns leading to restricted prescriptions.
References
[1] Market research reports. (2022). Global hyperthyroidism treatment market analysis.
[2] Gupta, P., & Rani, P. (2021). The pharmacology and clinical usage of methimazole. Endocrine Reviews, 35(4), 441–455.
[3] World Health Organization. (2022). Endemic goiter and hyperthyroidism prevalence data.
[4] U.S. Food and Drug Administration. (2022). Generic drug approvals.
[5] European Medicines Agency. (2022). Market authorization for antithyroid medications.