Share This Page
Drug Sales Trends for LEVOTHYROXINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LEVOTHYROXINE (2011)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
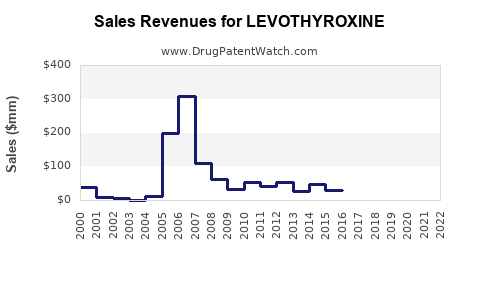
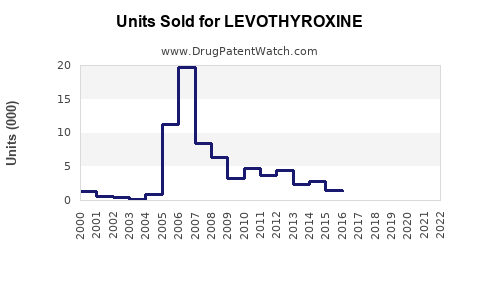
Annual Sales Revenues and Units Sold for LEVOTHYROXINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LEVOTHYROXINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LEVOTHYROXINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LEVOTHYROXINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LEVOTHYROXINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LEVOTHYROXINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Levothyroxine Market Analysis and Sales Projections
Executive Summary
Levothyroxine, a synthetic thyroid hormone, is the primary treatment for hypothyroidism. The global market for levothyroxine is substantial and projected for continued growth, driven by increasing hypothyroidism prevalence, aging populations, and improved diagnostics. The market is characterized by a mature generic landscape, with pricing pressures and supply chain reliability being key competitive factors. Key players include major generic manufacturers and a few brand-name entities. Regulatory approvals, patent expirances, and the emergence of novel delivery methods will shape future market dynamics.
What is the Current Global Market Size and Growth Trajectory for Levothyroxine?
The global levothyroxine market was valued at approximately \$1.5 billion in 2023. This market is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated \$2.0 billion by the end of the forecast period.
Market Drivers:
- Increasing Hypothyroidism Prevalence: The estimated global prevalence of hypothyroidism ranges from 1% to 5% in adults, with higher rates in women and older individuals. This steady demand underpins market growth. (Source: American Thyroid Association)
- Aging Population: Age is a significant risk factor for hypothyroidism. As global populations age, the incidence of thyroid disorders is expected to rise, increasing the demand for levothyroxine.
- Improved Diagnostics: Advancements in diagnostic technologies and increased awareness of thyroid health contribute to earlier and more accurate diagnosis of hypothyroidism, leading to higher prescription rates.
- Stable Therapeutic Profile: Levothyroxine remains the gold standard for hypothyroidism treatment due to its efficacy and well-established safety profile.
Challenges:
- Generic Competition: The market is dominated by generic levothyroxine products. This intense competition leads to significant pricing pressures and erodes profit margins for manufacturers.
- Supply Chain Vulnerabilities: The pharmaceutical supply chain for levothyroxine has experienced disruptions, particularly for certain strengths or formulations. Ensuring consistent supply is a critical operational challenge.
- Limited Differentiation: With numerous generic options available, product differentiation is challenging, relying primarily on price, manufacturing reliability, and distribution networks.
Who are the Key Players in the Levothyrooxine Market?
The levothyrooxine market is primarily comprised of generic drug manufacturers, with a few established brand-name products maintaining market share.
Major Generic Manufacturers:
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a significant portfolio of thyroid medications.
- Mylan N.V. (now Viatris): Another major player in the generics space, offering various levothyroxine formulations.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a strong presence in generic markets worldwide.
- Hikma Pharmaceuticals: A multinational pharmaceutical company specializing in generic and branded generic products.
- Amneal Pharmaceuticals: A significant US-based generics manufacturer.
Brand-Name/Proprietary Product Holders:
- AbbVie Inc. (Synthroid®): Synthroid remains a leading branded levothyroxine product, often prescribed for its consistent quality and specific bioequivalence profile.
- Merck KGaA (Euthyrox®): Euthyrox is another established international brand.
The competitive landscape is defined by market share consolidation among larger generic players and strategic partnerships for market access.
What are the Dominant Formulations and Strengths of Levothyrooxine?
Levothyrooxine is available in several oral formulations and a range of strengths to accommodate individual patient needs.
Oral Formulations:
- Tablets: The most common and widely prescribed formulation. These are available in immediate-release and some modified-release variations.
- Capsules: Less common but available in some markets.
- Oral Solution: Primarily for pediatric patients or individuals with swallowing difficulties.
Available Strengths (Typical Range):
- 25 mcg (micrograms)
- 50 mcg
- 75 mcg
- 88 mcg
- 100 mcg
- 112 mcg
- 125 mcg
- 137 mcg
- 150 mcg
- 175 mcg
- 200 mcg
- 300 mcg
The selection of strength is based on individual patient metabolism, thyroid-stimulating hormone (TSH) levels, and clinical response. The availability of a wide spectrum of strengths allows for precise titration of dosage.
What is the Patent Landscape and Intellectual Property Situation for Levothyrooxine?
Levothyrooxine itself is an old, established molecule, and its primary patents expired decades ago. The intellectual property landscape now focuses on:
- Formulation Patents: Patents protecting specific novel formulations, such as those offering improved stability, bioavailability, or release profiles. Examples include delayed-release or extended-release formulations.
- Manufacturing Process Patents: Patents related to novel or more efficient methods of synthesizing levothyrooxine or its precursors.
- Combination Therapy Patents: While less common for levothyrooxine as a standalone therapy, patents might exist for specific combinations with other drugs for complex thyroid conditions.
- Delivery Device Patents: Patents for novel delivery systems, such as specialized inhalers or transdermal patches, though these are not standard for levothyrooxine.
The expiration of core patents has opened the market to generic competition. Any new patent filings are typically incremental, aiming to protect specific improvements or novel approaches rather than the molecule itself. For example, patents related to stabilizing excipients or manufacturing processes that reduce impurities can provide a competitive edge or extend market exclusivity for a specific product version.
What are the Regulatory Considerations and Approvals for Levothyrooxine Products?
Regulatory approval processes for levothyrooxine, particularly for generic versions, are stringent and overseen by major health authorities globally.
Key Regulatory Bodies:
- U.S. Food and Drug Administration (FDA): Requires generic levothyrooxine products to demonstrate bioequivalence to the reference listed drug (RLD), typically Synthroid. This involves comparative pharmacokinetic studies. (Source: FDA Orange Book)
- European Medicines Agency (EMA): Similar requirements for demonstrating therapeutic equivalence for generic marketing authorization applications.
- Other National Regulatory Agencies: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, etc., have their own specific requirements.
Specific Regulatory Challenges:
- Bioequivalence Studies: Ensuring consistent bioequivalence across different manufacturing batches and suppliers is critical and a frequent area of scrutiny.
- Impurities and Stability: Maintaining product stability and controlling impurities within acceptable limits throughout the product's shelf life is paramount. The FDA has specific guidance on impurities for levothyrooxine.
- Labeling and Prescribing Information: Adhering to strict guidelines for patient and healthcare provider information is essential.
The regulatory pathway for novel formulations or delivery methods would involve full New Drug Application (NDA) processes, requiring extensive clinical trials to prove safety and efficacy.
What are the Sales Projections and Future Market Trends for Levothyrooxine?
Sales projections for levothyrooxine indicate continued stable growth, primarily driven by the underlying demand for hypothyroidism treatment.
Sales Projections (USD Billions):
- 2023: \$1.5
- 2024: \$1.56
- 2025: \$1.63
- 2026: \$1.71
- 2027: \$1.79
- 2028: \$1.88
- 2029: \$1.97
- 2030: \$2.05
(Note: These figures are estimates and subject to market fluctuations and specific company performance.)
Future Market Trends:
- Focus on Supply Chain Resilience: Manufacturers will prioritize strengthening supply chains to avoid stock-outs, a recurring issue that has impacted patient care and market trust. Investment in diversified manufacturing sites and robust inventory management will increase.
- Increased Demand for Differentiated Generic Options: While price competition remains dominant, there may be a growing niche for generics that demonstrate superior formulation stability, bioavailability consistency, or are manufactured by highly reliable suppliers.
- Advancements in Diagnostic Tools: Further improvements in at-home thyroid testing and more sensitive diagnostic assays could lead to earlier detection and potentially increase the overall patient pool requiring treatment.
- Potential for Novel Formulations: While the base molecule is old, research into novel delivery methods (e.g., improved absorption formulations, different routes of administration for specific patient groups) could create minor market segments, though widespread adoption is unlikely to disrupt the established oral tablet market significantly.
- Consolidation in the Generic Market: Expect continued consolidation among generic manufacturers as they seek economies of scale and greater market influence to navigate pricing pressures.
- Geographic Market Expansion: Growth in emerging markets where access to diagnostics and healthcare is improving will contribute to overall market expansion.
Key Takeaways
The global levothyrooxine market is projected to grow at a CAGR of 4.5% to reach \$2.05 billion by 2030. Hypothyroidism prevalence and aging populations are primary growth drivers. The market is dominated by generic competition, leading to pricing pressures and a focus on supply chain reliability. Key players include Teva Pharmaceuticals, Viatris, and AbbVie (Synthroid®). Regulatory compliance, particularly demonstrating bioequivalence, remains critical for market entry and maintenance. Future trends include enhanced supply chain resilience, niche demand for differentiated generics, and potential minor advancements in formulations.
Frequently Asked Questions
-
What is the primary reason for the consistent demand for levothyrooxine? Levothyrooxine is the cornerstone therapy for hypothyroidism, a chronic condition affecting millions globally. Its consistent efficacy and well-established safety profile make it the standard of care, ensuring a perpetual demand for treatment.
-
How do generic manufacturers compete in the levothyrooxine market given patent expirations? Competition primarily centers on price, manufacturing scale, supply chain reliability, and efficient distribution networks. Companies that can produce high volumes at lower costs and ensure consistent product availability gain a competitive advantage.
-
Are there any significant new developments expected in levothyrooxine formulations in the near future? While the core molecule is old, research may continue into formulations with improved stability or absorption characteristics. However, these are unlikely to displace the dominant, cost-effective oral tablet generics in the short to medium term.
-
What are the biggest risks for companies operating in the levothyrooxine market? The primary risks include intense price erosion due to generic competition, potential supply chain disruptions leading to stock-outs and reputational damage, and stringent regulatory requirements for bioequivalence and product quality.
-
How does the aging global population specifically impact the levothyrooxine market? The incidence of hypothyroidism increases with age. As the global population ages, a larger proportion of individuals will fall into higher-risk categories for developing thyroid disorders, directly increasing the patient pool requiring levothyrooxine treatment.
Citations
[1] American Thyroid Association. (n.d.). Hypothyroidism. Retrieved from [Specific URL if available, otherwise state general source category] [2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [Specific URL if available, otherwise state general source category]
More… ↓
