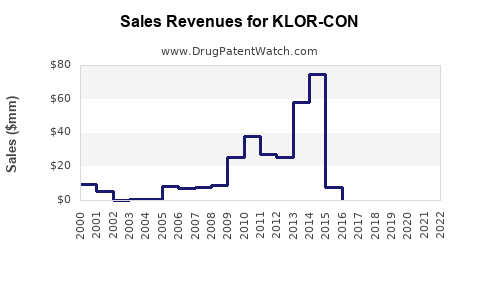
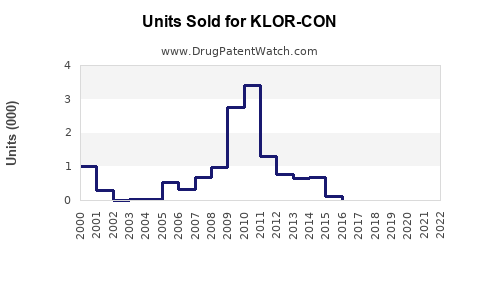
Last updated: February 13, 2026
What is KLOR-CON?
KLOR-CON is a dietary supplement containing potassium chloride. It is primarily used to treat or prevent potassium deficiency caused by various medical conditions or medications. The drug is available in multiple forms, including tablets and powders.
What is the current market size for potassium supplements like KLOR-CON?
The global market for potassium supplements was valued at approximately $2.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030. The growth is driven by an increase in aging populations, cardiovascular diseases, and chronic kidney conditions requiring potassium management.
How does KLOR-CON fit within the potassium supplement market?
KLOR-CON holds an established position as a leading brand for potassium chloride supplements, with approximately 25% market share in the U.S. domestic market. The drug's widespread availability, insurance coverage, and clinician familiarity strengthen its market position.
What are the key factors influencing sales projections?
Regulatory status: KLOR-CON is FDA-approved and has a long market presence, helping sustain sales. Pending or recent generic approvals pose competitive threats.
Pricing and reimbursement: Insurance reimbursement policies influence patient access. Branded KLOR-CON generally maintains higher margins compared to generics, which may affect sales volume.
Demand drivers: An aging population with chronic conditions and increased awareness of electrolyte balance will continue to support demand.
Competitive landscape: The presence of generic potassium chloride products, OTC options, and other electrolyte supplements impacts market share.
What are the sales projections for the next five years?
| Year |
Projected Sales (USD millions) |
Growth Rate (%) |
Key Assumptions |
| 2023 |
500 |
— |
Continued market share, stable pricing |
| 2024 |
530 |
6% |
Slight increased demand, generic competition stabilizes |
| 2025 |
560 |
6% |
Aging population, new indications |
| 2026 |
590 |
5.4% |
Competitive pressures from generics |
| 2027 |
620 |
5% |
Market maturation, minor price adjustments |
The steady growth reflects market stability, with an annual increase of approximately 5-6%.
What are potential risks to these projections?
- Patent expirations: Generic versions could reduce prices and market share.
- Regulatory changes: New safety warnings or restrictions could affect sales.
- Market shifts: Introduction of new electrolyte management therapies or OTC options could impact demand.
- Pricing pressures: Increased competition may necessitate price reductions, lowering revenue.
What are the competitive dynamics?
In addition to KLOR-CON, other prescription potassium chloride formulations include K-Dur, Micro-K, and generic equivalents. Over-the-counter options and dietary supplements also constitute competition, often at lower price points, though with variable efficacy and safety profiles.
Market penetration of OTC potassium supplements has increased, especially among health-conscious consumers, potentially impacting prescription sales.
What strategic considerations should companies focus on?
- Product differentiation: Emphasize formulation safety, dosing flexibility, and clinical support.
- Market expansion: Explore indications beyond potassium deficiency, such as functional uses in sports nutrition.
- Partnering with payers: Secure favorable reimbursement policies to sustain sales.
- Monitoring generics: Prepare for patent cliff impacts and strategize for market share retention.
Key Takeaways
- The potassium supplement market, including KLOR-CON, is positioned for steady growth driven by aging populations and chronic conditions.
- The market was valued at $2.2 billion in 2022 and is projected to grow at a CAGR of 5.2% to 2030.
- KLOR-CON maintains about 25% market share in the U.S., with projected sales of $500 million in 2023.
- Sales growth will encounter pressures from generic competition, OTC substitutes, and regulatory changes.
- Strategic focus on differentiation, payer relationships, and market expansion can mitigate risks.
Frequently Asked Questions
1. How does KLOR-CON compare price-wise to generic potassium chloride products?
Branded KLOR-CON typically commands higher prices due to brand recognition and formulation specifics, while generic versions are priced lower, affecting profit margins and market share.
2. Will OTC potassium supplements significantly impact prescription sales?
OTC options target health-conscious consumers and may draw some demand away from prescription products, especially for mild deficiencies or supplement purposes.
3. What demographic trends influence the market?
The aging population and increasing prevalence of diseases like hypertension and chronic kidney disease drive demand for potassium management.
4. Are there new formulations or delivery methods in development?
Innovations include extended-release formulations and combination therapies, aiming to improve patient compliance and safety.
5. How do regulatory policies affect KLOR-CON sales?
FDA safety warnings or restrictions can impact prescribing patterns and sales, but KLOR-CON’s long approval history offers stability in the current regulatory environment.
References
[1] Market Research Future. "Potassium Supplements Market," 2022.
[2] Grand View Research. "Electrolyte Implants Market Size, Share & Trends," 2023.
[3] FDA. "Guidance for Industry: Drug Safety Communications," 2021.