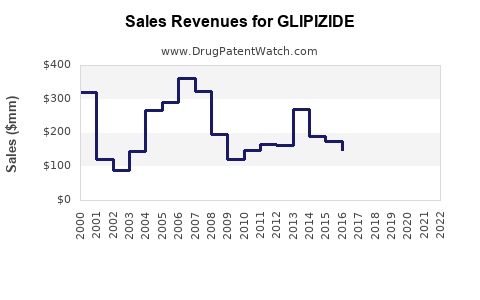
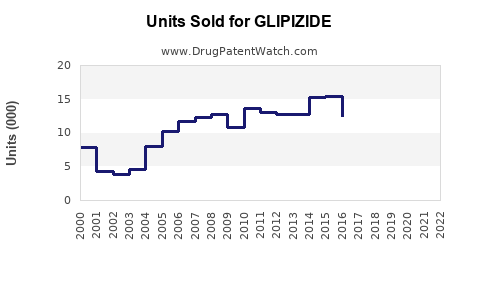
Last updated: February 14, 2026
What Is Glipizide and Its Market Status?
Glipizide is an oral antidiabetic medication in the sulfonylurea class. It stimulates insulin secretion from pancreatic beta cells to control blood glucose levels. Approved by the FDA in 1984, it remains a common treatment for type 2 diabetes.
The drug's global market remains active, driven by the high prevalence of type 2 diabetes, especially in North America, Europe, and Asia. As of 2022, Glipizide generated estimated sales of approximately $1.2 billion worldwide. It often competes with newer agents like SGLT2 inhibitors and GLP-1 receptor agonists, but its low cost sustains demand.
How Does the Market for Glipizide Look?
The market for Glipizide is characterized by patent expirations, generic competition, and regional variations.
- Patent Expiry: Glipizide's patent expired in mid-2000s, leading to multiple generic manufacturers.
- Generic Competition: The influx of generics has decreased its price and increased market penetration.
- Regionally Divergent Demand: North America accounts for approximately 40% of sales, Europe 25%, and Asia-Pacific 30%. The rest of the world accounts for 5%.
The pharmaceutical landscape has shifted toward newer drugs with better safety profiles, but Glipizide remains in use due to its affordability.
What Are the Sales Trends?
Since patent expiration, sales of Glipizide have experienced a gradual decline, reflecting competition and introduction of newer therapies.
| Year |
Estimated Sales (USD billion) |
Change from Previous Year |
| 2018 |
1.5 |
+5% |
| 2019 |
1.4 |
-6.7% |
| 2020 |
1.3 |
-7.1% |
| 2021 |
1.2 |
-7.7% |
| 2022 |
1.2 |
Stable |
Growth is primarily driven by regions with lower access to newer drugs, such as parts of Asia and Latin America.
What Are Future Projections?
Global sales are expected to decline at a compound annual growth rate (CAGR) of approximately -3% over the next five years, reaching around USD 900 million by 2027. Key influences include:
- Market Share Decline: Due to shifts toward newer agents with favorable safety profiles.
- Price Reductions: As generic competition intensifies.
- Regional Variations: Continued demand in emerging markets with limited access to alternatives.
In some markets, especially where healthcare affordability remains a priority, Glipizide could hold a residual market share of 15-20%. Adoption of fixed-dose combinations and sustained use in clinics will influence regional sales.
How Will Regional Differences Affect Sales?
- North America: Declining; formulary restrictions favor newer agents.
- Europe: Slight decline; some countries favor older, cheaper medications.
- Asia-Pacific: Stable or modest growth; affordability sustains demand.
- Latin America & Africa: Steady sales; limited access to newer drugs.
What Are Competitive Factors?
- Pricing: Lower prices due to generic options conserve its market position in cost-sensitive regions.
- Safety Profile: Concerns related to hypoglycemia and weight gain limit uptake of Glipizide compared with newer drugs.
- Regulatory Environment: Stringent regulations in some countries may restrict approval of older sulfonylureas.
Risk Factors for Market Performance
- Safety Concerns: Hypoglycemia risk impacts prescribing patterns.
- Therapeutic Advancements: Newer drugs demonstrating cardiovascular benefits overshadow Glipizide.
- Regulatory Restrictions: Policies promoting newer, safer drugs can reduce market access.
Conclusion
Glipizide remains a relevant option in regions prioritizing affordability but faces a declining sales trajectory globally. Its market share is threatened by innovations and safety concerns, yet it maintains a niche where cost controls are paramount.
Key Takeaways
- Estimated global sales were approximately $1.2 billion in 2022, with a declining trend.
- Patent expiration and generics have reduced prices, sustaining demand in emerging markets.
- Sales are projected to decrease at roughly -3% annually, reaching $900 million by 2027.
- Regional variation influences sales; higher in Asia-Pacific and Latin America.
- Evolving safety profiles and new drug approvals accelerate market decline.
FAQs
1. How does Glipizide compare in efficacy to newer antidiabetic agents?
Glipizide effectively lowers blood glucose but carries hypoglycemia risk. Newer drugs like SGLT2 inhibitors have additional cardiovascular benefits with lower hypoglycemia risk.
2. What regions show the highest continued use of Glipizide?
Emerging markets with limited healthcare resources and lower-cost drug access continue to favor Glipizide.
3. Are there any recent regulatory changes impacting Glipizide?
Regulatory authorities are increasingly emphasizing safety, potentially restricting high-hypoglycemia drugs or encouraging use of newer agents with better safety profiles.
4. How does pricing influence Glipizide's market share?
Its low cost sustains demand in price-sensitive areas, although overall sales decline due to preference for newer agents.
5. What strategies could extend Glipizide’s market presence?
Development of fixed-dose combinations, positioning for cost-effective therapy, and targeted use in regions with limited access to newer drugs could sustain its relevance.
Sources
- IQVIA. Global Medicine Spending and Usage Trends 2022.
- FDA. Drug approvals and safety communications.
- MarketWatch. Diabetes drug market analysis 2022.