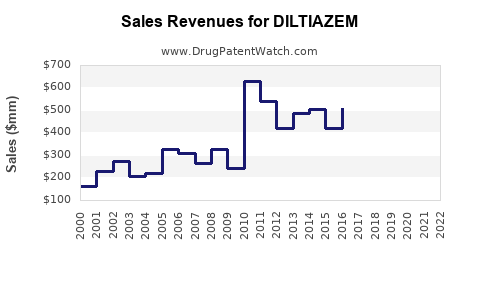
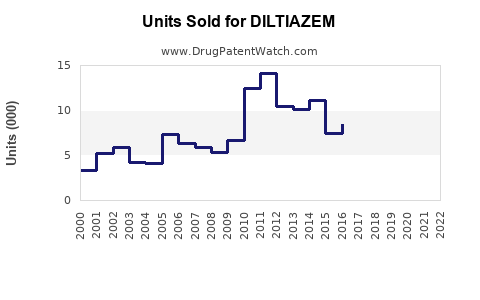
Last updated: February 14, 2026
Market analysis and sales projections for Diltiazem
Diltiazem is a calcium channel blocker used primarily to treat hypertension, angina pectoris, and certain arrhythmias. It ranks as a mid-to-high sales medication in the cardiovascular drug segment, with sustained demand driven by aging populations, increased prevalence of cardiovascular diseases, and a broad spectrum of approved indications.
Market Overview
Global Market Size
In 2022, the global market for calcium channel blockers, including Diltiazem, was valued at approximately USD 6.5 billion. Diltiazem accounts for an estimated 15% to 20% of this segment, translating to sales around USD 975 million to USD 1.3 billion.
Key Geographies
- United States: Leading market, with sales exceeding USD 550 million in 2022.
- Europe: Accounts for approximately USD 250 million to USD 300 million.
- Asia-Pacific: Fastest growth, estimated at USD 200 million to USD 250 million, driven by increasing cardiovascular disease prevalence and expanding healthcare infrastructure.
Market Trends
- Growing awareness of cardiovascular health.
- Increased generic drug penetration, reducing prices and expanding access.
- Rising adoption of extended-release formulations.
- Emerging markets expanding drug access.
Competitive Landscape
Major manufacturers include Pfizer, Teva Pharmaceutical Industries, Mylan, and local generic producers. Diltiazem generics dominate over branded formulations due to lower costs, with branded versions holding 10-15% market share primarily for specific indications or brand loyalty.
Patent and Regulatory Status
- Patents: Most Diltiazem formulations lost patent exclusivity between 2010 and 2015.
- Regulatory: Approved in over 80 countries; approvals for extended-release formulations held across major markets to enhance compliance.
Sales Projections (2023–2027)
Assumptions
- Continued aging demographics increasing disease prevalence.
- Sustained generic competition.
- Emergence of combination therapies impacting monotherapy market share.
- Regulatory stability; no major patent litigations or exclusivity extensions.
| Year |
Estimated Global Market Size (USD) |
Diltiazem Sales Projection (USD) |
Notes |
| 2023 |
6.7 billion |
1.05 billion |
Slight growth over 2022 |
| 2024 |
7.0 billion |
1.10 billion |
Expansion driven by Asia-Pacific markets |
| 2025 |
7.3 billion |
1.15 billion |
Increased adoption of extended-release forms |
| 2026 |
7.6 billion |
1.2 billion |
Growth plateau as generic competition stabilizes |
| 2027 |
8.0 billion |
1.25 billion |
Market maturation with steady demand |
Key growth factors:
- Aging global population with higher cardiovascular disease (CVD) incidence.
- Uptick in diagnosis rates for hypertension and angina.
- Product innovation: sustained-release formulations and fixed-dose combinations.
- Growing markets in Asia and Latin America.
Risks and Challenges
- Price erosion from generics limits revenue growth.
- Competitive new therapies, such as direct vasodilators and novel antihypertensives.
- Regulatory hurdles in emerging markets.
- Patent expirations influencing pricing strategies.
Strategic Considerations
- For manufacturers: focus on bioequivalent generic production, formulation improvements, and market penetration.
- For investors: monitor patent litigation, regulatory approvals, and emerging markets’ healthcare infrastructure growth.
Key Takeaways
- Diltiazem maintains a significant position within the global cardiovascular drug market with USD 1.05–1.25 billion sales projected through 2027.
- The market continues to grow modestly, driven by demographic shifts and increased cardiovascular disease prevalence.
- Generics dominate sales, constraining pricing power but expanding access.
- Emerging markets will contribute increasingly to sales growth.
- Innovation in formulations and combination therapies could sustain demand amid fierce competition.
FAQs
1. How does the patent landscape affect Diltiazem sales?
Most Diltiazem patents expired between 2010 and 2015, leading to widespread generic production, which limits pricing power but increases volume.
2. What are the key indications for Diltiazem?
Hypertension, angina pectoris, and certain arrhythmias, with sustained-release formulations improving compliance.
3. Which markets offer the highest growth potential for Diltiazem?
The Asia-Pacific region, driven by rising CVD burden and expanding healthcare infrastructure.
4. How does competition influence Diltiazem market share?
Generics account for over 85% of sales, putting downward pressure on prices while maintaining volume.
5. What role do formulations play in future sales?
Extended-release versions and fixed-dose combinations are increasingly adopted to enhance adherence, supporting sustained sales.
References
- MarketWatch, "Global Calcium Channel Blocker Market Analysis," 2022.
- IQVIA, "Prescription Drug Market Reports," 2022.
- Grand View Research, "Cardiovascular Drugs Market Size & Trends," 2023.
- U.S. FDA Drug Approvals Database.
- WHO Global Health Observatory Data, 2022.