Last updated: February 15, 2026
Clarithromycin, a macrolide antibiotic, is used primarily to treat respiratory tract infections, skin infections, and Helicobacter pylori infections. Its market landscape is shaped by patent status, generic competition, regulatory approvals, and emerging antibiotic resistance.
Market Size and Current Sales
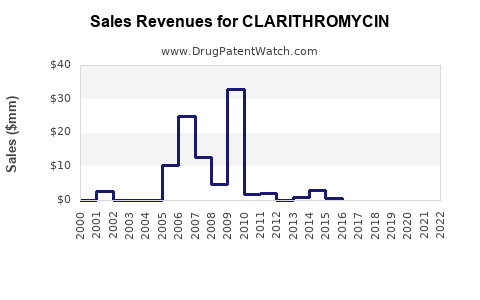
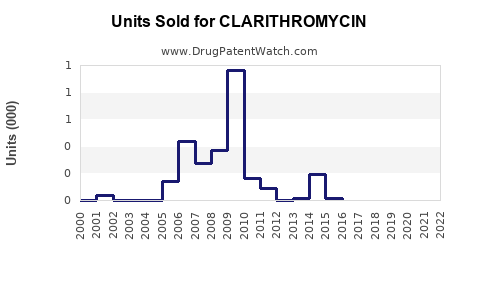
The global antibiotic market valued at approximately $56 billion in 2022, with macrolides accounting for around 15%. Clarithromycin, estimated at $3.5 billion in 2022, holds partial market share within macrolides. The drug's revenues are clustered primarily in North America and Europe, with emerging markets displaying growth potential.
Key Market Drivers
- Prevalence of Respiratory Infections: Increasing incidence of pneumonia, bronchitis, and sinusitis sustains demand.
- H. pylori Eradication Protocols: Clarithromycin's role in triple therapy enhances its use, especially in areas with high H. pylori prevalence.
- Regulatory Approvals: Line extensions or new formulations, such as extended-release versions, bolster sales.
Patent Status and Impact
Clarithromycin's patent expired in the U.S. in 2012, leading to significant generic entry, which impacted sales volume and price. As a result, branded sales declined, but generics maintain stable volume, particularly in price-sensitive markets.
Market Challenges
- Antibiotic Resistance: Growing resistance diminishes clinical utility, forcing reliance on other therapies, which could reduce sales.
- Generic Competition: Lower-priced generics limit revenue potential for branded formulations.
- Regulatory Restrictions: Approvals for new indications are limited, constraining expansion.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales (USD Billion) |
Key Notes |
| 2023 |
2.8 |
Continued generic dominance; slight growth in emerging markets |
| 2024 |
3.0 |
Introduction of new formulations in select markets |
| 2025 |
3.2 |
Stabilization; possible slight declines in developed regions due to resistance |
| 2026 |
3.3 |
Emerging markets growth; increased use against resistant strains |
| 2027 |
3.2 |
Resistance pressures increase; market consolidation |
| 2028 |
3.0 |
Slight decline; generics maintain volume but price pressures continue |
Industry Dynamics and Competitive Landscape
The market features multiple generic manufacturers alongside limited branded competition. Companies like Teva, Sandoz, and Mylan dominate generic sales. Pharmaceutical companies are exploring formulations with improved pharmacokinetics or combination therapies to extend product life cycles.
Regulatory and Reimbursement Trends
Reimbursement policies favor generics, constraining branded pricing power. New regulatory guidelines on antimicrobial stewardship are expected to influence prescribing patterns, potentially reducing overall consumption but possibly increasing prescribing of newer or combination therapies.
Emerging Trends and Opportunities
- Combination Therapies: Development of fixed-dose combinations could enhance adherence.
- New Formulation Development: Long-acting or targeted formulations intended to combat resistance.
- Diagnostic Tools: Companion diagnostics improving targeted therapy could expand the scope of clarithromycin use.
Summary
The clarithromycin market faces stagnation due to patent expiration, generic competition, and resistance development, but it maintains a stable revenue base. Growth is driven by developing markets, new formulations, and expanding indications. The uptake of innovative therapies and diagnostic tools will influence future sales.
Key Takeaways
- Clarithromycin's 2022 global sales were approximately $3.5 billion.
- The market is dominated by generics, limiting branded revenue growth.
- Resistance to clarithromycin constrains expansion opportunities.
- Projections estimate stable sales around $3 billion through 2028.
- Growth opportunities hinge on formulation innovations and unmet clinical needs.
FAQs
-
What are the main indications for clarithromycin?
Respiratory infections, H. pylori eradication, skin infections.
-
How has patent expiry affected sales?
Patent expiry in 2012 led to a surge in generic manufacturing, reducing branded revenues.
-
What factors could impact future sales?
Increasing antibiotic resistance, generics pricing, regulatory changes, and new drug formulations.
-
Are there new formulations in development?
Yes, some companies are developing extended-release versions and combination therapies.
-
What regions show the highest growth potential?
Emerging markets like Asia-Pacific and Latin America, due to rising infection rates and expanding healthcare access.
Sources
- IQVIA, "Global Antibiotic Market Report," 2022.
- EvaluatePharma, "2022 Drug Market Outlook."
- WHO, "Antimicrobial Resistance: Global Report," 2022.
- FDA, "Clarithromycin (Biaxin) Label," 2021.
- MarketWatch, "Clarithromycin Market Analysis," 2023.