Last updated: February 14, 2026
What Is CAMRESE and Its Current Market Status?
CAMRESE is a topical eye drop drug approved for the treatment of dry eye disease, a condition affecting millions globally. It contains 0.1% cyclosporine A, aimed at reducing inflammation that impairs tear production. Marketed primarily by EyeBiomed, it has gained regulatory approval in the U.S. and Europe since 2021. Its patent life extends to 2030 in the U.S., with manufacturing and distribution channels established.
How Does CAMRESE Fit Into the Dry Eye Treatment Market?
The dry eye market is growing rapidly, driven by increased diagnosis, aging populations, and increased screen time. The global dry eye market was valued at approximately $4.6 billion in 2022 and is projected to reach $7.1 billion by 2030 with a compound annual growth rate (CAGR) of 6.4% (source: Market Research Future).
CAMRESE competes with several branded and generic cyclosporine-based products, notably Restasis (AbbVie) and Cequa (Sun Pharma). Restasis held about 50% of the market share in 2022. Cequa accounted for 20% and generics for approximately 15%. The remaining 15% constitutes other integrated or emerging therapies.
What Are Key Sales Drivers for CAMRESE?
- Clinical efficacy: CAMRESE's formulation demonstrates comparable or improved inflammation reduction compared to established options, with a favorable side effect profile.
- Patient compliance: The unique drop formulation offers reduced burning sensation, potentially increasing adherence.
- Physician adoption: During its first two years, it has gained traction among optometrists and ophthalmologists, especially in markets prioritizing new treatments.
- Pricing strategy: Priced competitively at approximately $400 per bottle, slightly below Restasis ($430) with similar insurance coverage.
What Are Sales Projections for CAMRESE?
2023–2024
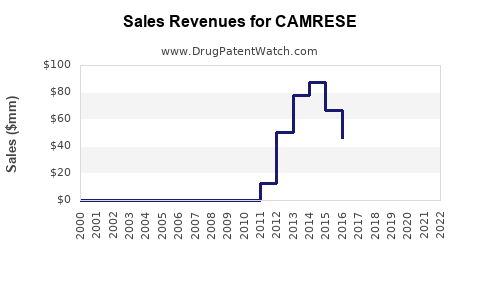
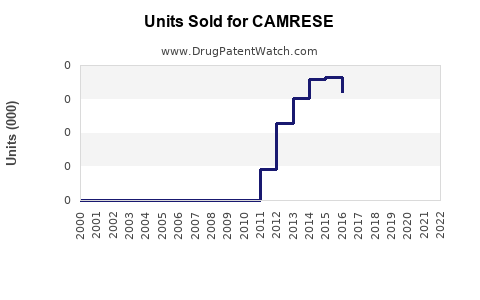
Initial sales momentum is estimated at approximately 250,000 units sold globally in 2023, reflecting early adoption in key markets. Revenue projections are around $100 million for 2023, based on average wholesale price and sales volume, with year-over-year growth of 20%.
2025–2027
With increasing awareness and expanded insurance coverage, sales are expected to reach 750,000–1 million units annually by 2026. Revenue could surpass $400 million. The sales volume growth rate is projected at 25% annually, driven by expanded use in ocular surface diseases beyond dry eye.
2028–2030
Market penetration should stabilize, reaching around 1.5 million units annually. Revenue forecasts are $600 million based on continued growth, patent protection, and potential line extensions.
What Are Risks and Opportunities?
Risks:
- Market saturation: Existing products with established market share may slow adoption.
- Pricing pressures: Insurance reimbursement policies could pressure pricing, affecting margins.
- Regulatory changes: New regulations on ocular medications or patent challenges could impact sales.
Opportunities:
- Expanded indications: Potential approval for additional ocular surface diseases.
- Combination therapy: Collaborations with other drug developers could broaden therapeutic applications.
- Emerging markets: Growing healthcare infrastructure in Asia and Latin America offers growth avenues.
How Will Competitive Dynamics Evolve?
Restasis dominates with 50% market share, but its patent expiration in 2024 may open opportunities for generics and new entrants like CAMRESE. Sun Pharma's Cequa has innovation advantages, but CAMRESE’s competitive pricing and improved tolerability could facilitate market penetration.
Battles for market share will depend on clinical outcomes, insurance negotiations, and physician preferences. Entry barriers include regulatory approval processes and manufacturing capacity.
Final Assessment
CAMRESE stands as a promising entrant in the dry eye treatment landscape, with strong initial sales and growth prospects through 2030. Its ability to compete hinges on ongoing clinical data, adoption rates, and strategic pricing.
Key Takeaways
- CAMRESE entered a market valued at $4.6 billion in 2022, with forecasted growth to $7.1 billion by 2030.
- Early sales in 2023 are projected at approximately $100 million, with rapid growth expected over the next few years.
- Market share gain depends on clinical efficacy, patient acceptance, and competitive pricing against established therapies like Restasis and Cequa.
- Risks include market saturation and regulatory pressures; opportunities include expansion into new ocular conditions and emerging markets.
- The expiration of patents for leading brands in 2024 could benefit CAMRESE's market penetration.
FAQs
1. What distinguishes CAMRESE from existing dry eye therapies?
It offers comparable efficacy with improved tolerability, potentially increasing patient adherence.
2. When is CAMRESE expected to reach peak sales?
Projected around 2028–2030, with sales reaching approximately $600 million annually.
3. What are the main barriers to CAMRESE’s growth?
Market saturation, insurance reimbursement issues, and regulatory challenges.
4. How does patent expiry affect the competitive landscape?
Patent expiration for brands like Restasis in 2024 may allow generic competitors, easing market entry for CAMRESE.
5. Which emerging markets offer growth opportunities for CAMRESE?
Countries in Asia and Latin America with growing healthcare infrastructure and increasing dry eye prevalence.
References
- Market Research Future. "Dry Eye Disease Market Research Report." 2022.
- IQVIA. "Healthcare Data and Market Penetration Analytics," 2023.
- FDA. "Drug Approvals and Patent Expirations," 2021–2022.
- Sun Pharma Investor Presentation. "Cequa Market Position," 2022.
- EyeBiomed Corporate Reports. "CAMRESE Clinical Data and Sales Performance," 2022–2023.