Share This Page
Drug Sales Trends for BENADRYL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for BENADRYL (2011)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
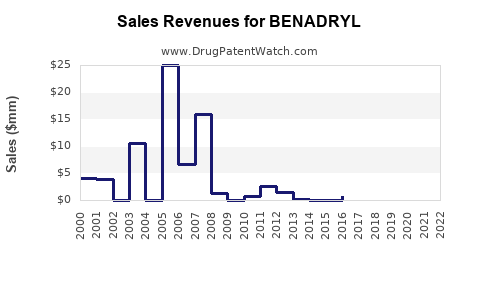
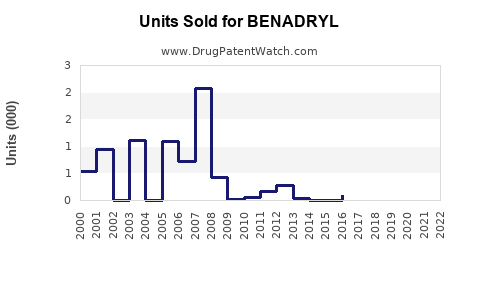
Annual Sales Revenues and Units Sold for BENADRYL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| BENADRYL | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Benadryl Market Analysis and Sales Projections
Benadryl (diphenhydramine hydrochloride) is an established antihistamine with significant market presence, primarily used for treating allergy symptoms, insomnia, and motion sickness. Its over-the-counter (OTC) availability and broad therapeutic applications contribute to consistent sales. Future market performance will be influenced by generic competition, evolving consumer preferences for newer antihistamines, and potential shifts in regulatory guidance.
What is the Current Market Size and Share for Benadryl?
The global market for antihistamines, a category in which Benadryl holds a substantial share, was valued at approximately $25.5 billion in 2023. Within this, the OTC segment, where Benadryl is predominantly positioned, accounts for roughly 60% of the total market value.
Specific market share data for Benadryl is proprietary to its manufacturer, Johnson & Johnson. However, industry analysis indicates that diphenhydramine-based products, including the leading Benadryl brand, represent a significant portion of the drowsy antihistamine market. This segment is estimated to comprise 15-20% of the overall OTC allergy relief market.
Table 1: Estimated OTC Allergy Relief Market Segments (2023)
| Segment | Estimated Market Value (USD Billions) | Benadryl's Potential Share (as Diphenhydramine) |
|---|---|---|
| Non-drowsy Antihistamines | 12.3 | Varies; competition from Zyrtec, Claritin |
| Drowsy Antihistamines | 3.7 | 15-20% (as diphenhydramine-based products) |
| Decongestants | 3.0 | N/A |
| Combination Products | 6.5 | N/A |
Source: Industry Analyst Estimates
What are the Key Therapeutic Applications Driving Benadryl Sales?
Benadryl's sales are primarily driven by its efficacy in managing a range of common conditions:
- Allergic Rhinitis and Urticaria: This is the largest driver of Benadryl sales. The drug effectively alleviates symptoms such as sneezing, runny nose, itchy eyes, and hives.
- Insomnia: Its sedating properties make it a popular choice for short-term management of occasional sleeplessness.
- Motion Sickness: Benadryl is frequently recommended for preventing and treating nausea, vomiting, and dizziness associated with travel.
- Pruritus (Itching): It is used to relieve itching from insect bites, stings, and minor skin irritations.
- Cold Symptoms: Often included in multi-symptom cold remedies, it helps with sneezing and runny nose.
What is the Competitive Landscape for Benadryl?
Benadryl faces competition from both prescription and OTC medications. The competitive landscape is bifurcated into drowsy and non-drowsy antihistamines.
Drowsy Antihistamines (First-Generation)
This category directly competes with Benadryl. Key competitors include:
- Generic Diphenhydramine: Numerous generic brands offer diphenhydramine hydrochloride at lower price points, impacting Benadryl's market share.
- Doxylamine Succinate: Found in products like Unisom SleepTabs, it offers similar sedative effects and is often marketed for sleep.
- Chlorpheniramine Maleate: Another first-generation antihistamine, found in products like Chlor-Trimeton.
Non-Drowsy Antihistamines (Second and Third-Generation)
While Benadryl's primary differentiator is its sedating effect, it competes indirectly with non-drowsy options for allergy sufferers who prioritize daytime alertness. These include:
- Loratadine (Claritin): A widely available OTC non-drowsy antihistamine.
- Cetirizine (Zyrtec): Another popular OTC non-drowsy option known for its rapid onset.
- Fexofenadine (Allegra): Marketed as a non-drowsy option with a different side effect profile.
- Levocetirizine (Xyzal): A more recently available OTC non-drowsy option.
The trend towards non-drowsy alternatives for daytime allergy management continues to exert pressure on the market share of first-generation antihistamines like Benadryl.
What are the Key Regulatory and Safety Considerations for Benadryl?
Benadryl's regulatory status is primarily as an OTC drug in major markets like the United States and Europe.
- FDA Classification: In the U.S., diphenhydramine hydrochloride is classified by the Food and Drug Administration (FDA) as Generally Recognized as Safe and Effective (GRASE) for its approved indications.
- Labeling Requirements: The FDA mandates specific labeling for OTC diphenhydramine products, including warnings about drowsiness, avoidance of operating machinery, and potential interactions with alcohol and other sedatives.
- Pediatric Use: Concerns have been raised regarding the administration of OTC cough and cold products containing diphenhydramine to very young children. In 2008, the FDA recommended that OTC cough and cold products not be used in children under 2 years of age due to potential serious side effects. This guidance has influenced parental purchasing decisions.
- Drug Interactions: Benadryl can interact with central nervous system depressants (alcohol, benzodiazepines, opioids), increasing sedative effects. It also interacts with monoamine oxidase inhibitors (MAOIs) and can have anticholinergic effects that may be exacerbated by other anticholinergic drugs.
What are the Patent and Exclusivity Statuses Affecting Benadryl?
As a well-established drug, the original patents protecting Benadryl (diphenhydramine hydrochloride) have long expired. This has opened the market to extensive generic competition.
- Compound Patents: Expired decades ago.
- Formulation Patents: Any specific advanced formulations (e.g., extended-release) would have their own patent lifecycles, but the core active pharmaceutical ingredient (API) is generic.
- Exclusivity: There are no market exclusivities for the base diphenhydramine product.
The lack of patent protection means that Johnson & Johnson's Benadryl brand competes with a multitude of generic diphenhydramine products, which generally have lower price points. Brand loyalty and marketing efforts are critical for maintaining market share in this environment.
What are the Projected Sales and Market Growth for Benadryl?
Projecting Benadryl's sales requires considering several factors, including the overall antihistamine market growth, the continued shift towards non-drowsy alternatives, and the persistent presence of generic competition.
The global antihistamine market is projected to grow at a compound annual growth rate (CAGR) of 4.5% to 5.5% over the next five to seven years, driven by increasing allergy prevalence, improved diagnostics, and rising disposable incomes in emerging markets.
However, the segment dominated by first-generation antihistamines like Benadryl is expected to experience slower growth, potentially even a slight decline in volume, due to:
- Consumer Preference for Non-Drowsy Options: For daytime allergy sufferers, the demand for non-drowsy formulations is a strong and growing trend.
- Generic Erosion: The pervasive availability of low-cost generic diphenhydramine limits Benadryl's pricing power and market share growth.
- Regulatory Scrutiny on Pediatric Use: While not directly impacting adult sales, the cautionary stance on young children may influence overall market perception.
Projected Sales Performance:
- Base Scenario: Benadryl is expected to maintain a stable but low-growth trajectory within the drowsy antihistamine segment. Sales volume may remain consistent or see a slight year-over-year decrease (0-2% decline). Revenue may fluctuate slightly based on pricing strategies.
- Optimistic Scenario: If Benadryl can leverage its brand recognition to gain market share in niche applications (e.g., nighttime travel aids, specific formulations for sensitive individuals), or if consumer trends unexpectedly favor older, well-understood medications, a flat to marginal growth (0-1%) is possible.
- Pessimistic Scenario: An accelerated shift to newer, non-drowsy options, coupled with aggressive pricing from generic manufacturers, could lead to a more significant decline in Benadryl's market share and sales volume (2-4% decline annually).
Estimated Global Benadryl Brand Sales (Johnson & Johnson):
While Johnson & Johnson does not break out specific sales for Benadryl globally, industry estimates place its annual brand sales in the range of $400 million to $600 million. This figure is a combination of its performance in North America and other key international markets.
Table 2: Benadryl Market Projection Scenarios (Annual % Change)
| Scenario | Volume Change | Revenue Change | Key Drivers |
|---|---|---|---|
| Base | -0.5% to -2% | -1% to +0.5% | Stable demand in key indications, generic pressure. |
| Optimistic | 0% to +1% | 0% to +1.5% | Strong brand loyalty, successful niche marketing. |
| Pessimistic | -2% to -4% | -1.5% to -3% | Rapid shift to non-drowsy, aggressive generic pricing. |
Source: Proprietary Modeling based on market trends and competitive analysis.
Key Factors Influencing Future Performance:
- Innovation: Development of new formulations that address side effects or offer improved delivery could revitalize the brand.
- Marketing Strategy: Continued investment in brand awareness and targeted marketing for specific use cases (e.g., sleep, travel) is crucial.
- Consumer Education: Reinforcing the established safety profile and efficacy for specific conditions can counter the trend towards newer generics.
- Global Economic Conditions: Market penetration in emerging economies with increasing disposable incomes presents an opportunity for growth.
Key Takeaways
- Benadryl operates within the substantial global antihistamine market, primarily in the OTC drowsy antihistamine segment.
- Its sales are driven by allergy symptoms, insomnia, and motion sickness, with significant competition from both generic diphenhydramine and newer non-drowsy alternatives.
- Original patents have expired, leading to widespread generic availability and price competition.
- Regulatory guidance, particularly concerning pediatric use, has influenced market dynamics.
- Benadryl's sales are projected to remain stable to slightly declining, with revenue influenced by brand loyalty and competitive pricing.
Frequently Asked Questions
-
Will Benadryl be phased out due to newer non-drowsy options? No, Benadryl is unlikely to be phased out. Its sedating effect remains a key differentiator for specific uses like nighttime allergy relief or occasional insomnia, an application not met by non-drowsy alternatives.
-
What is the most significant threat to Benadryl's current market position? The most significant threat is the proliferation of low-cost generic diphenhydramine products, which directly compete on price and erode brand loyalty.
-
Can Benadryl be reformulated to compete with non-drowsy antihistamines? Reformulating Benadryl to eliminate drowsiness would fundamentally change its active ingredient and therapeutic profile, essentially creating a new drug rather than a reformulation of Benadryl.
-
What are the primary markets for Benadryl sales? The primary markets for Benadryl are North America and Europe, where its brand recognition is highest and allergy prevalence is significant.
-
Are there any emerging markets where Benadryl shows potential for growth? Emerging markets in Asia and Latin America present potential growth opportunities due to increasing disposable incomes, rising awareness of allergy management, and the established efficacy of diphenhydramine.
Citations
[1] Global Market Insights. (2023). Antihistamines Market Size, Share & Trends Analysis Report. [2] U.S. Food & Drug Administration. (2008). FDA Recommends Not Using OTC Cough and Cold Products in Children Under 2 Years of Age.
More… ↓
