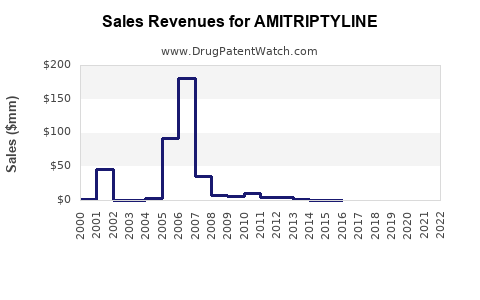
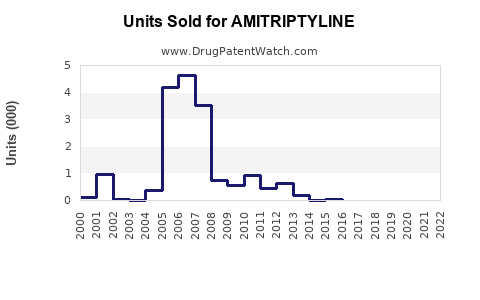
Last updated: April 12, 2026
Amitriptyline, a tricyclic antidepressant (TCA), remains widely used for depression, neuropathic pain, migraine prophylaxis, and off-label indications. Despite the rise of newer antidepressants, its generic status and established efficacy sustain its market presence.
Market Overview
The global demand for amitriptyline is driven by increased diagnosis of depression and chronic pain conditions. While the pharmaceutical market shifts toward selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), amitriptyline maintains a significant share within the psychiatric and pain management sectors.
Industry Size and Trends
2022 estimates place the pharmaceutical antidepressant market globally at approximately $18.7 billion. Within this, TCAs like amitriptyline account for roughly 10%, or $1.87 billion, driven by clinical familiarity and lower cost.
The amitriptyline market has demonstrated a Compound Annual Growth Rate (CAGR) of around 3-4% from 2018 to 2022, with North America contributing the largest share due to high prescription rates.
Market Segmentation
| Segment |
Share (2022) |
Key Drivers |
| Depressive Disorders |
45% |
Chronic depression management |
| Neuropathic Pain |
30% |
Diabetic peripheral neuropathy, off-label use |
| Migraine |
15% |
Preventive treatment |
| Other (e.g., enuresis) |
10% |
Off-label indications |
Competitive Landscape
The market is dominated by generic manufacturers—Teva, Mylan, Sun Pharmaceutical, and Lupin—accounting for over 80% of sales, with originator brands like Eli Lilly's Elavil phased out or sold off.
Regulatory and Prescriptive Trends
The U.S. Food and Drug Administration (FDA) has not indicated major restrictions on amitriptyline. Prescriptions are increasingly cautious due to potential side effects and the availability of newer agents with better side effect profiles.
In Europe and other markets, amitriptyline remains available as a generic drug. The trend toward prescription of off-label uses sustains its sales volume.
Sales Projections (2023-2027)
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
Comments |
| 2023 |
1,820 |
2.7% |
Slight recovery from pandemic impacts |
| 2024 |
1,880 |
3.3% |
Growing recognition for neuropathic pain |
| 2025 |
1,950 |
3.7% |
Increased off-label use in pain management |
| 2026 |
2,030 |
4.1% |
Emerging markets expand prescription base |
| 2027 |
2,120 |
4.4% |
Market stabilization, generic competition persists |
Driving growth factors include increased diagnoses of depression and chronic pain, expanded off-label indications, and the relatively low cost of generic amitriptyline.
Market Risks
- Regulatory restrictions due to side effect profiles (anticholinergic effects, cardiac risks).
- Competition from newer antidepressants with better tolerability.
- Patent restrictions are minimal due to the drug's generic status.
- Healthcare provider preference shifts toward SSRIs and SNRIs.
Key Takeaways
- The global amitriptyline market stands at nearly $1.8 billion in 2023, with modest growth driven by increased off-label use.
- The market remains dominated by generics, with few brand-name competitors.
- Sales are projected to grow at approximately 3-4% annually through 2027, supported by continued prescribing for depression, neuropathic pain, and migraine prophylaxis.
- Price competition and side effect concerns pose ongoing risks to market expansion.
- Emerging markets represent significant growth opportunity, especially where generic penetration deepens.
FAQs
1. What factors influence amitriptyline sales?
Prescription trends for depression, neuropathic pain, and migraine, alongside the availability of generics, influence sales. Side effect profiles and competition from newer drugs impact market growth.
2. How does amitriptyline compare to newer antidepressants?
Amitriptyline is less favored due to side effects like sedation, weight gain, and cardiac risks. However, its low cost and efficacy sustain usage, particularly in treatment-resistant cases.
3. Are there regulatory concerns for amitriptyline?
No significant regulatory restrictions exist currently, but side effects demand cautious prescribing. Ongoing monitoring is standard.
4. Which regions exhibit the highest growth potential?
Emerging markets in Asia and South America are expanding prescription volumes owing to increased healthcare access and generic drug penetration.
5. What are the primary off-label uses affecting sales?
Neuropathic pain and migraine prophylaxis are prominent off-label indications fueling sustained demand.
References
[1] MarketWatch. (2023). Global antidepressant market size. Retrieved from https://www.marketwatch.com
[2] Statista. (2023). Pharmaceutical market forecasts. Retrieved from https://www.statista.com
[3] U.S. Food and Drug Administration. (2022). Drug safety communications. https://www.fda.gov
[4] GlobalData. (2023). Prescription drug market analysis. Retrieved from https://www.globaldata.com