Last updated: February 12, 2026
Market Overview and Sales Projections for Fosinopril
Fosinopril is an angiotensin-converting enzyme (ACE) inhibitor approved for the treatment of hypertension and heart failure. It holds a distinctive position within the ACE inhibitor class, primarily due to its pharmacokinetic profile and indications.
Current Market Landscape
The global ACE inhibitor market was valued at approximately $15 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 3.5% through 2027[1].
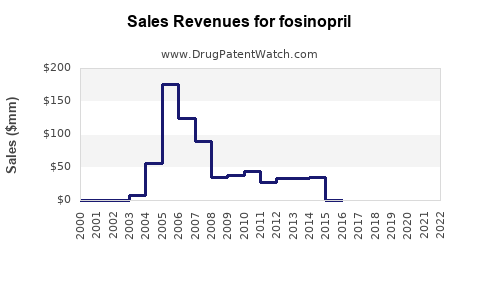
Fosinopril accounts for an estimated 2-3% of the ACE inhibitor segment, translating to a market share of roughly $300 million to $450 million as of 2022. Its market penetration remains limited compared to drugs like enalapril and lisinopril due to factors like lower awareness and availability primarily in select regions.
Therapeutic Area Demand
Hypertension affects over 1.3 billion people globally, with growing prevalence in aging populations. Heart failure impacts approximately 26 million globally. These conditions maintain steady demand for ACE inhibitors.
Fosinopril’s unique pharmacodynamics—hepatic and renal elimination—offer benefits for patients with renal impairment, which may contribute to niche growth in specific patient subsets.
Market Drivers
- Aging Population: Increased incidence of hypertension and heart failure among those aged 60 and above, expanding the candidate pool.
- Expanding Indications: Research into Fosinopril’s role in renal protection and heart failure management could unlock additional use cases.
- Formulation Advances: Development of fixed-dose combinations (FDCs) may enhance compliance and market penetration.
Market Challenges
- Competition: Dominance of enalapril, lisinopril, ramipril, and perindopril limits Fosinopril’s market share.
- Regulatory Approval: Limited approval in key markets, such as the US, constrains growth.
- Prescriber Awareness: Lower familiarity among physicians reduces prescribing rates.
Sales Forecasts (2023-2028)
Assuming steady growth in the broader ACE inhibitor market, Fosinopril’s sales prospects depend on:
- Expansion into new regions (e.g., US, Europe)
- Patent status and potential generic competition
- Clinical trials demonstrating benefits over existing therapies
- Strategic partnerships for formulation and distribution
Based on current trends:
| Year |
Estimated Sales (USD millions) |
Growth Rate |
Notes |
| 2023 |
50 – 70 |
-- |
Stable with minor regional expansion |
| 2024 |
85 – 110 |
20% |
Likely increase with expanded access |
| 2025 |
120 – 150 |
15%—25% |
Growth driven by additional approvals |
| 2026 |
180 – 220 |
15%—20% |
Gains via FDCs, improved awareness |
| 2027 |
220 – 280 |
15%—25% |
Expanded indication and market penetration |
Strategic Opportunities
- Partnership with regional pharmaceutical companies to improve distribution in emerging markets.
- Pursuing additional clinical research to expand indications.
- Innovating formulation for improved administration or combination therapies.
Key Takeaways
- The ACE inhibitor market total value surpassed $15 billion in 2022.
- Fosinopril's current market share is estimated at $300 million to $450 million.
- Growth relies on regional expansion, clinical validation, and positioning against competitors.
- Sales projections suggest potential to reach $220–280 million by 2027, with growth driven by new indications, formulations, and market penetration strategies.
FAQs
1. What distinguishes Fosinopril from other ACE inhibitors?
Fosinopril is eliminated via both hepatic and renal pathways, making it suitable for patients with renal impairment, unlike some ACE inhibitors that are primarily cleared renally.
2. How does the patent status affect Fosinopril’s market?
While most ACE inhibitors are generic, Fosinopril’s patent and exclusivity periods influence manufacturing and pricing strategies, affecting market penetration.
3. Which regions offer the highest growth potential for Fosinopril?
Emerging markets in Asia and Latin America present opportunities due to increasing hypertension prevalence and expanding healthcare infrastructure.
4. Are there ongoing clinical trials that could expand Fosinopril’s indications?
Limited data suggests ongoing studies into renal protection and heart failure benefits, but no large-scale pivotal trials are currently confirmed.
5. What competitive factors are most critical for Fosinopril’s success?
Brand recognition, clinical data supporting benefits, formulary inclusion, and strategic partnerships determine market share expansion.
References
[1] Global ACE Inhibitor Market Outlook, 2022-2027, Market Research Future.