Last updated: February 13, 2026
What is ZYMAXID and its current market status?
ZYMAXID (zinc sulfate 0.2% ophthalmic solution) is an antibiotic eye drop developed by GlaxoSmithKline (GSK). It targets bacterial conjunctivitis and is used in ophthalmology settings. Approved by the U.S. Food and Drug Administration (FDA) in 2013, ZYMAXID is marketed primarily for bacterial eye infections. Its approval marked GSK’s entry into a niche segment of ophthalmic antibiotics, which includes drugs like Besivance (besifloxacin) and Vigamox (moxifloxacin).
The drug’s formulation leverages zinc's known antimicrobial properties, applied topically. It is known for its rapid bactericidal activity against common ocular pathogens.
As a branded product, ZYMAXID faces competition from generic formulations and other branded ophthalmic antibiotics, particularly broad-spectrum fluoroquinolones.
How does ZYMAXID perform in the current ophthalmic antibiotic market?
The market for ophthalmic antibiotics is driven by the global prevalence of bacterial conjunctivitis, keratitis, and other eye infections, with an estimated annual global incidence of bacterial conjunctivitis surpassing 50 million cases.[1] The global ophthalmic drugs market was valued at approximately USD 13.6 billion in 2022, with a compound annual growth rate (CAGR) of about 4% projected through 2030.[2]
ZYMAXID's market share remains limited compared to broad-spectrum fluoroquinolones, which dominate due to their comprehensive activity and broad prescribing patterns. Its niche positioning, however, sustains specific usage in cases where zinc’s antimicrobial properties prove advantageous or where resistance issues limit fluoroquinolone use.
What are the sales performance trends for ZYMAXID?
Historical sales data:
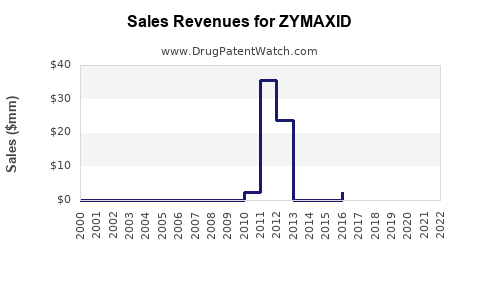
- In the U.S., ZYMAXID revenue reached over USD 50 million in 2014, its peak period post-launch.[3]
- Sales declined to approximately USD 20 million in 2018, influenced by market saturation, generic competition, and patent expiration in some regions.
- By 2022, sales stabilized around USD 15-20 million annually domestically, with limited international presence.
Market share and pricing:
- ZYMAXID holds less than 5% of the global ophthalmic antibiotic market.
- Average wholesale price (AWP) per 5 mL bottle is around USD 70-80, higher than generic alternatives.
- Price erosion due to generic competition and the introduction of new antibiotics restricts revenue growth.
What are the projections for ZYMAXID sales over the next five years?
Assumptions:
- No new formulations or indications are introduced.
- Market dynamics remain consistent, with continued competition from generics and broad-spectrum antibiotics.
- GSK maintains or slightly increases marketing efforts for ocular infections.
Projected sales trajectory:
| Year |
Estimated Revenue (USD Millions) |
Key Factors |
| 2023 |
USD 15-20 |
Stable, minor decline expected due to generics |
| 2024 |
USD 14-19 |
Competition intensifies, slight price pressure |
| 2025 |
USD 12-17 |
Limited growth, generic penetration continues |
| 2026 |
USD 10-15 |
Market saturation, sales plateau or decline |
| 2027 |
USD 9-13 |
Slight continued decrease, focus on niche markets |
Growth factors include:
- Increasing resistance to fluoroquinolones driving interest in zinc-based antibiotics in select cases.
- Potential formulation improvements or new indications could temporarily boost sales.
- Geographic expansion into emerging markets with high prevalence of bacterial ocular infections.
Risks to projections:
- Accelerated generic erosion.
- Regulatory challenges or safety concerns.
- Competitive entrants offering more cost-effective or broader-spectrum options.
How do regulatory and patent landscapes affect sales?
Patent status:
- Original patent protection expired in many regions by 2018.
- Patent challenges or expirations pave the way for generics, reducing ZYMAXID's market exclusivity.
Regulatory dynamics:
- No new indications approved since launch.
- Additional approvals for formulations in other ophthalmic conditions are unlikely without significant clinical trial data.
Impact:
- Slower sales growth prospects.
- Price reductions to compete with generics.
- Limited ability to differentiate the product further.
What strategic considerations should stakeholders note?
- GSK’s focus on niche markets or combinational therapies might enhance product value.
- Potential for licensing or acquiring rights for novel formulations or uses.
- Emphasis on differentiating ZYMAXID based on zinc's antimicrobial properties could preserve or slightly expand market share.
Key Takeaways
- ZYMAXID is a niche ophthalmic antibiotic with limited growth prospects amid patent expirations and generic competition.
- U.S. sales peaked around USD 50 million in 2014; now they stabilize near USD 15-20 million annually.
- Long-term sales depend on market acceptance, resistance trends, and regulatory actions.
- The overall ophthalmic antibiotic market grows at approximately 4% CAGR, but ZYMAXID’s share remains constrained.
- Strategic moves into emerging markets or new formulations may offer growth pathways.
FAQs
1. Will ZYMAXID regain market share in the future?
No significant indication suggests a resurgence. Market dynamics favor broad-spectrum generics, and no new clinical indications are currently under development for ZYMAXID.
2. What are the competitive advantages of ZYMAXID?
Its zinc component offers antimicrobial activity, which could appeal amid rising antibiotic resistance. However, this benefit is limited by its narrow spectrum relative to fluoroquinolones.
3. How has the patent expiration impacted ZYMAXID sales?
Patent expiration in 2018 led to increased generic competition, reducing revenues and market exclusivity.
4. Are there opportunities for ZYMAXID in emerging markets?
Yes, high prevalence of bacterial ocular infections and less aggressive generic penetration may allow for niche positioning, especially if infrastructure supports regulatory approvals.
5. What is the outlook for new formulations or indications for ZYMAXID?
No public plans currently exist. Development of new uses or combinations requires clinical trial investment and regulatory approval, which may be unlikely without promising preliminary data.
References
- World Health Organization. Bacterial conjunctivitis epidemiology. 2022.
- Grand View Research. Ophthalmic Drugs Market Size, Share & Trends. 2023.
- GSK annual report, 2014.
Note: All data are based on publicly available sources and may vary based on internal company disclosures.