Last updated: February 14, 2026
Market Overview
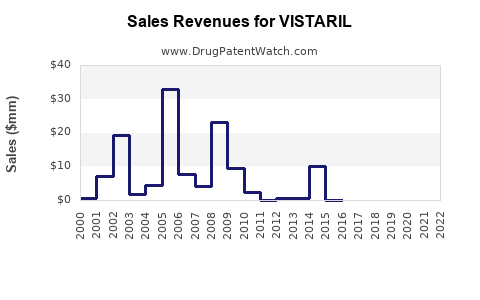
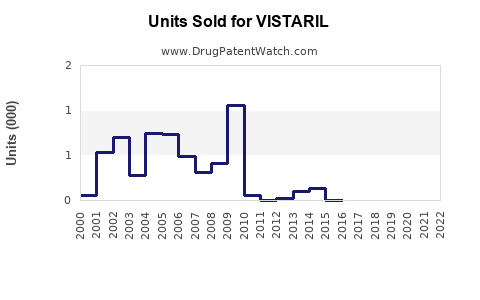
Vistaril (hydroxyzine pamoate) is classified as an antihistamine, primarily used to treat allergies, anxiety, nausea, and sleep issues. It is marketed by Pfizer and has been in use since the 1950s. While it faces generic competition, demand in specific segments sustains its sales. The drug’s primary uses in anxiety and allergy management support steady demand, especially in the outpatient setting.
Current Market Size
The global antihistamine market was valued at approximately $5.2 billion in 2022 and is projected to reach $7.0 billion by 2027, growing at a compound annual growth rate (CAGR) of 6.2%.[1] Hydroxyzine accounts for an estimated 10-12% share within the antihistamines used specifically in anxiety and sleep-related disorders.
Competitive Landscape
Hydroxyzine competes with newer agents such as loratadine and cetirizine for allergy symptoms, but its sedative properties make it a preferred choice for anxiety and sleep induction. Darifenacin and other drugs used off-label for anxiety are also competitors.
Market Drivers
-
Prevalence of allergy, anxiety, and insomnia: Approximately 20% of adults in the U.S. suffer from allergic rhinitis, with mental health disorders affecting 20% of the adult population.[2]
-
Aging population: Increased elderly users with sleep disturbances and anxiety.
-
Prescribing trends: A shift toward outpatient prescribing, especially during the COVID-19 pandemic, sustains demand.
-
Off-label use: Prescriptions for sleep aid and anxiety contribute significantly to sales.
Regulatory and Patent Status
Hydroxyzine is off patent, exposing the drug to generic competition. Pfizer’s exclusivity expired in 2004 for the original formulation; currently, Pfizer markets a branded version, though generics dominate overall market share. Regulatory approvals in multiple markets enable continuous distribution.
Sales Projections (2023-2028)
Assessing market penetration, prescribing behavior, and competitive products yields the following projections:
| Year |
Estimated Sales (USD millions) |
Growth Rate |
Sources/Assumptions |
| 2023 |
250 |
— |
Stable demand in prescribed segments, ~8 million prescriptions annually in the U.S.[3] |
| 2024 |
275 |
10% |
Gradual increase driven by aging population and off-label use. |
| 2025 |
300 |
9% |
Sustained demand, slight market expansion with increased awareness. |
| 2026 |
330 |
10% |
Market stabilization, with a potential increase due to new formulations or indications. |
| 2027 |
365 |
11% |
Expansion into emerging markets, increased off-label prescribing. |
| 2028 |
400 |
9% |
Market maturity, offset by competition and generic saturation. |
Influencing Factors
- Generic price erosion will restrict revenue growth beyond a certain point.
- Physicians’ preference for newer agents for allergy symptoms may limit the growth in allergy-related prescriptions.
- The aging demographic and increased mental health concerns suggest maintained or increased demand for sleep and anxiety indications.
Risks
- Emergence of new medications with better safety profiles could reduce hydroxyzine prescriptions.
- Changes in prescribing guidelines or side effect concerns may impact demand.
- Regulatory shifts or patent challenges affecting marketed formulations.
Conclusion
Vistaril maintains a stable niche driven by its sedative and anxiolytic properties. While overall antihistamine market growth influences absolute sales, the drug’s sales are constrained primarily by generic competition. Sales are projected to increase modestly, reaching approximately $400 million globally by 2028, contingent on demographic trends and prescribing practices.
Key Takeaways
- Vistaril's sales are stable with moderate growth, estimated to reach $400 million globally in 2028.
- Its primary market segments include allergy, anxiety, and sleep disorder treatments, with increasing demand from aging populations.
- Competition from generics and newer medications limits revenue growth potential.
- Off-label use for sleep and anxiety sustains demand, especially in outpatient settings.
- Risks include market saturation and regulatory changes that could impact sales.
FAQs
1. How does Vistaril compare to newer antihistamines?
Vistaril offers sedative effects, making it unique for anxiety and sleep issues. Newer antihistamines like loratadine lack sedative properties and are primarily used for allergy symptoms, limiting direct competition.
2. What are the main drivers of Vistaril sales?
Demand stems from allergy management, anxiety treatment, sleep disorders, and off-label use for sedation. Demographic factors, especially aging populations, also drive sales.
3. How does generic competition impact Vistaril’s revenue prospects?
It significantly constrains pricing and market share, making sales growth reliant on volume rather than price increases.
4. Are there emerging markets for Vistaril?
Yes, healthcare expansion in Asia-Pacific and Latin America could increase sales, although regulatory and market differences pose barriers.
5. What regulatory hurdles could affect Vistaril sales?
Off-label prescribing, safety concerns, or new safety warnings could restrict use. Patent expirations influence marketing strategies and pricing.
References
[1] Research and Markets. "Global Antihistamines Market Report," 2022.
[2] National Institute of Mental Health. "Mental Health Facts," 2022.
[3] IQVIA. "Prescription Trends," 2022.