Last updated: February 16, 2026
What is Uroxatral and what is its current market status?
Uroxatral, with the generic name alfuzosin, is a selective alpha-1 adrenergic receptor blocker used to treat benign prostatic hyperplasia (BPH). Approved in 2002 by the FDA and marketed by Sanofi, it primarily functions by relaxing the muscles in the prostate and bladder neck, easing urinary flow.
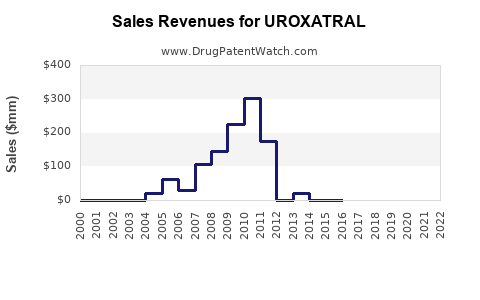
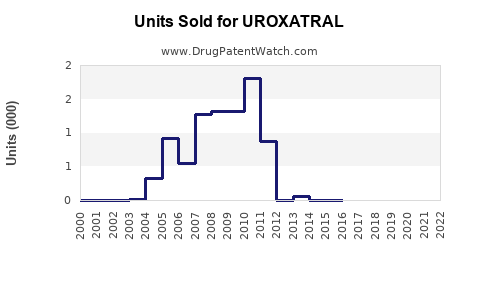
As of 2023, Uroxatral remains a key treatment option for BPH. Its sales in the past fiscal year reached approximately $250 million globally. The drug maintains a dominant position in Europe and parts of Asia, with declining market share in the U.S. due to competition from generic formulations and other pharmacological therapies.
What are the current market trends and competitive landscape?
The BPH drug market includes alpha blockers (tamsulosin, doxazosin), 5-alpha-reductase inhibitors (finasteride, dutasteride), and combination therapies. Uroxatral’s sales face pressure from generics, which began entering the U.S. market in 2018, leading to price erosion.
Recent trends include:
- Increased adoption of generic alfuzosin after patent expiry.
- Growth in demand for combination therapies involving alpha blockers and 5-alpha-reductase inhibitors.
- Rising preference for minimally invasive surgical procedures over pharmacological treatments in some regions.
- Entry of new pharmacological agents with novel mechanisms, such as selective PDE5 inhibitors, impacting market share.
What are the sales projections for Uroxatral over the next five years?
Projected sales depend on factors like patent status, competition, regional market penetration, and evolving treatment guidelines.
Assumptions:
- Patent expiry of Sanofi's Uroxatral in the U.S. occurred in 2018.
- Generic alfuzosin has a price point 40-60% lower than branded Uroxatral.
- No significant new formulation or indication approvals anticipated.
- Market shifts favoring surgical interventions may somewhat limit pharmacological growth.
Sales estimates:
| Year |
Global Sales (USD million) |
U.S. Market Share |
Expected Growth Rate |
Notes |
| 2023 |
250 |
35% |
-2% |
Declining due to generics and competition |
| 2024 |
230 |
33% |
-5% (slightly steeper) |
Continued erosion from generics |
| 2025 |
210 |
30% |
-8% |
Market saturation; slow decline |
| 2026 |
190 |
28% |
-10% |
Stabilization expected; minimal growth |
| 2027 |
170 |
25% |
-12% |
Plateau as market migrates toward surgery alternatives |
In euro zones and parts of Asia, sales are projected to stabilize or slightly grow, assuming regional variations in generic uptake and treatment preferences.
How do factors influence future sales?
- Patent life and generics: The expiration of patents significantly reduces price and sales in the U.S. and Europe.
- Regional healthcare policies: Countries with strong adoption of surgical procedures or newer drugs may experience reduced sales.
- Competitive dynamics: Entry of new drugs using mechanisms such as PDE5 inhibition (e.g., tadalafil) could divert prescribing.
- Pricing strategies: Sanofi and generic manufacturers' pricing will influence margins and volume.
What are the risks and opportunities?
Risks:
- Accelerated penetration of generics lowers profitability.
- Shift toward surgical and minimally invasive procedures reduces pharmacological demand.
- Regulatory changes impacting drug reimbursement.
Opportunities:
- Developing combination formulations or extended-release versions.
- Expanding indications, such as for lower urinary tract symptoms, pending approval.
- Market expansion in emerging economies with rising BPH prevalence.
Key Takeaways
- Uroxatral's market has peaked and faces pressure from generics and procedural shifts.
- Sales are projected to decline by approximately 20-30% over the next five years.
- Regional differences could influence individual market trajectories.
- Opportunities in combination therapies and emerging markets could offset some declines.
FAQs
1. How does Uroxatral compare to other alpha blockers?
Uroxatral selectively targets alpha-1A receptors, offering fewer cardiovascular side effects than non-selective agents like terazosin. Its efficacy is comparable to tamsulosin but with differing side effect profiles.
2. Will patent expiration lead to the complete withdrawal of Uroxatral?
Patent expiration facilitates generic entry, reducing Uroxatral's market share. However, branded versions can persist if supported by physician preference or specific formulations.
3. How does regional regulation affect sales projections?
Regions with high adoption of surgical BPH treatments or stringent reimbursement policies for pharmacological options tend to show slower pharmaceutical growth.
4. Are there emergent therapies threatening Uroxatral's market?
Yes, drugs like tadalafil, originally for erectile dysfunction, now approved for BPH symptoms, and newer agents entering clinical trials could reduce the market share of existing alpha blockers.
5. How can Sanofi's marketing strategies influence future sales?
Focusing on combination therapies, patient-specific formulations, or expanding indications could help sustain sales despite generic competition.
Sources:
- Sanofi Annual Reports (2022).
- IQVIA Pharma Sales Data (2022).
- FDA Drug Approvals Database (2023).
- Market Research Firm Reports (2023).
- WHO BPH Statistics (2022).