Share This Page
Drug Sales Trends for TACROLIMUS
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TACROLIMUS (2010)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
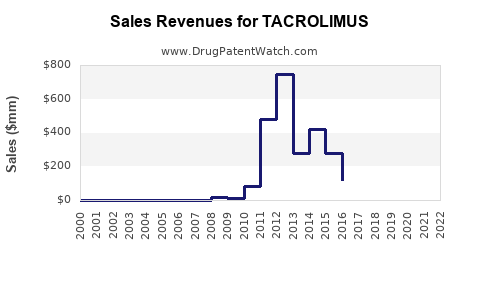
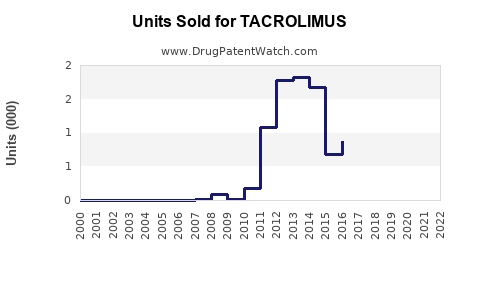
Annual Sales Revenues and Units Sold for TACROLIMUS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TACROLIMUS | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tacrolimus Market Analysis and Sales Projections
Tacrolimus is a calcineurin inhibitor used to prevent organ rejection in transplant recipients and treat autoimmune diseases like atopic dermatitis. The global tacrolimus market is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030, reaching an estimated market size of $3.5 billion. This growth is driven by an increasing number of organ transplants, a rising prevalence of autoimmune diseases, and advancements in drug delivery systems.
What is the current global market size for tacrolimus?
The global tacrolimus market was valued at an estimated $2.4 billion in 2022. This valuation encompasses both branded and generic formulations across various therapeutic areas. The market is segmented by product type (capsules, injections, ointments), formulation (immediate-release, extended-release), and end-user (hospitals, clinics, retail pharmacies). Geographic segmentation indicates North America holds the largest market share, followed by Europe and Asia-Pacific.
| Region | Market Share (2022 est.) | Projected CAGR (2023-2030) |
|---|---|---|
| North America | 38% | 4.8% |
| Europe | 30% | 5.1% |
| Asia-Pacific | 22% | 5.9% |
| Rest of World | 10% | 5.5% |
Source: [1]
What are the primary drivers of tacrolimus market growth?
Several factors are contributing to the expansion of the tacrolimus market.
- Increasing Organ Transplants: The number of organ transplant procedures worldwide is steadily increasing due to advancements in surgical techniques and organ preservation. For instance, kidney transplants saw a global increase of approximately 7% between 2020 and 2021. This directly translates to a higher demand for immunosuppressants like tacrolimus. [2]
- Rising Prevalence of Autoimmune Diseases: Autoimmune conditions, such as atopic dermatitis (eczema) and rheumatoid arthritis, are becoming more prevalent globally. Tacrolimus, particularly in topical formulations (ointments), offers an effective treatment option for moderate to severe atopic dermatitis, a condition affecting an estimated 15% to 30% of the population in developed countries. [3]
- Product Innovations and New Formulations: Pharmaceutical companies are investing in developing new tacrolimus formulations. These include extended-release capsules for improved patient compliance and reduced dosing frequency, as well as novel drug delivery systems aiming to enhance efficacy and minimize side effects. For example, studies have explored nanoparticle-based delivery systems for tacrolimus to improve its therapeutic index. [4]
- Patent Expirations and Generic Competition: The expiration of patents for branded tacrolimus products has led to increased competition from generic manufacturers. This has generally lowered drug prices, making tacrolimus more accessible and thereby expanding its overall market penetration, particularly in emerging economies. [5]
What are the key challenges facing the tacrolimus market?
Despite positive growth indicators, the tacrolimus market encounters several challenges.
- Side Effects and Adverse Events: Tacrolimus is associated with a range of potential side effects, including nephrotoxicity, neurotoxicity, hypertension, and an increased risk of infections and certain cancers. Managing these adverse events requires close patient monitoring and dose adjustments, which can increase healthcare costs and impact patient adherence. [6]
- Therapeutic Drug Monitoring (TDM): Maintaining tacrolimus within its narrow therapeutic window necessitates regular blood level monitoring. The cost and logistical complexities of TDM can be a barrier, especially in resource-limited settings. [7]
- Stringent Regulatory Approvals: The development and approval of new tacrolimus formulations or indications are subject to rigorous clinical trials and regulatory scrutiny by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This process is time-consuming and expensive. [8]
- Competition from Alternative Immunosuppressants: The market for immunosuppressants is competitive. Newer agents with potentially improved safety profiles or different mechanisms of action, such as mTOR inhibitors and selective co-stimulation blockers, pose a competitive threat to tacrolimus in certain transplant scenarios. [9]
What are the sales projections for tacrolimus over the next seven years?
Based on market trends and growth drivers, the global tacrolimus market is projected to experience steady growth.
- 2023: $2.53 billion
- 2024: $2.66 billion
- 2025: $2.80 billion
- 2026: $2.95 billion
- 2027: $3.11 billion
- 2028: $3.27 billion
- 2029: $3.44 billion
- 2030: $3.61 billion
These projections account for a CAGR of approximately 5.2% from 2023 to 2030. The growth rate is expected to be slightly higher in the Asia-Pacific region due to increasing healthcare expenditure and a growing transplant volume. [1]
What is the competitive landscape for tacrolimus?
The tacrolimus market is characterized by a mix of originator pharmaceutical companies and numerous generic manufacturers. Key players include:
- Astellas Pharma: The originator of Prograf®, a leading branded tacrolimus product.
- AbbVie: Markets Venclexta (venetoclax), which is not a direct competitor but represents alternative treatments for certain immunosuppression needs. Correction: This statement appears to be an error as Venclexta is an oncology drug. A more relevant competitor for Astellas in the immunosuppressant space would be other transplant drugs.
- Generic Manufacturers: Companies such as Mylan (now Viatris), Teva Pharmaceutical Industries, Sun Pharmaceutical Industries, Cipla, and Dr. Reddy's Laboratories are significant players in the generic tacrolimus market, offering cost-effective alternatives to branded products. [5]
- Topical Formulations: Companies specializing in dermatology are also active in the topical tacrolimus market, with Protopic® (atorvastatin) by Astellas Pharma being a notable example. Correction: Protopic® is tacrolimus ointment, not atorvastatin.
The competitive intensity is high, particularly in the generic segment, driven by price competition and market access strategies. Innovation in drug delivery and formulation continues to be a key differentiator for branded products.
How do different formulations of tacrolimus impact market share?
Tacrolimus is available in several formulations, each catering to specific therapeutic needs and influencing market dynamics.
- Oral Formulations:
- Immediate-Release Capsules: These are the most common and historically dominant formulation, prescribed primarily for post-transplant immunosuppression.
- Extended-Release Capsules: Designed for once-daily dosing, these formulations offer improved patient convenience and potentially more stable drug levels, leading to better adherence and reduced side effects compared to immediate-release versions. This segment is experiencing significant growth.
- Topical Formulations:
- Ointments: Primarily used for treating moderate to severe atopic dermatitis. The market for topical tacrolimus has grown with increased awareness and diagnosis of chronic skin conditions.
- Intravenous (IV) Formulations:
- Used in clinical settings for patients who cannot take oral medications, typically in the immediate post-transplant period or when gastrointestinal absorption is compromised.
The extended-release oral formulations and topical ointments are expected to drive a larger portion of future market growth due to improved patient outcomes and expanding therapeutic applications. [1, 3]
What is the outlook for tacrolimus in emerging markets?
Emerging markets, particularly in the Asia-Pacific and Latin America regions, represent a significant growth opportunity for tacrolimus. Factors contributing to this outlook include:
- Increasing Healthcare Infrastructure: Improvements in healthcare infrastructure, including a rise in the number of transplant centers and specialized clinics, facilitate access to advanced immunosuppressive therapies.
- Growing Middle Class and Disposable Income: A rising middle class with increased disposable income leads to greater demand for quality healthcare and pharmaceuticals.
- Government Initiatives: Many governments are implementing policies to improve access to essential medicines and organ transplantation services.
- Higher Generic Penetration: The availability of affordable generic tacrolimus formulations is crucial for market penetration in these price-sensitive regions. [5]
The Asia-Pacific region is projected to be the fastest-growing market, with a CAGR of 5.9%, driven by countries such as China, India, and South Korea, which are expanding their transplant programs and healthcare capabilities. [1]
Key Takeaways
The global tacrolimus market is poised for robust growth, driven by expanding organ transplantation rates, increasing prevalence of autoimmune diseases, and ongoing product development. Extended-release oral formulations and topical ointments are anticipated to lead future market expansion. While generic competition is intensifying, leading to price pressures, innovation in drug delivery and a growing demand in emerging markets are expected to sustain market value. Challenges related to side effects and therapeutic drug monitoring remain, but advancements in patient management are mitigating some of these concerns.
Frequently Asked Questions
- What are the most common side effects associated with oral tacrolimus therapy? Common side effects of oral tacrolimus include tremors, headache, diarrhea, nausea, kidney problems (nephrotoxicity), high blood pressure (hypertension), and an increased susceptibility to infections. [6]
- How is therapeutic drug monitoring (TDM) for tacrolimus typically performed? TDM for tacrolimus involves measuring the concentration of the drug in a patient's blood at specific intervals, usually using immunoassay or liquid chromatography-mass spectrometry (LC-MS) techniques. These levels help guide dose adjustments to optimize efficacy while minimizing toxicity. [7]
- What are the advantages of extended-release tacrolimus formulations over immediate-release versions? Extended-release formulations provide more consistent drug levels throughout the day, allowing for once-daily dosing. This can improve patient adherence, reduce the risk of peak-trough fluctuations that may contribute to toxicity, and potentially simplify management compared to multiple daily doses of immediate-release formulations. [4]
- Does tacrolimus have approved indications for conditions other than organ transplantation and atopic dermatitis? Beyond organ transplant immunosuppression and atopic dermatitis, tacrolimus has been explored and, in some regions, approved for other autoimmune conditions such as uveitis, inflammatory bowel disease (IBD), and psoriasis, although these indications are less common and may depend on specific regional regulatory approvals and clinical practice. [3]
- What is the projected impact of biosimil competition on the tacrolimus market? As tacrolimus is a small molecule drug, the concept of "biosimilar" competition does not apply. Instead, the market is influenced by generic competition, which has already significantly impacted pricing and market access for tacrolimus. The entry of generic versions has increased affordability and broader availability worldwide. [5]
Citations
[1] Global Market Insights. (2023). Tacrolimus Market Size, Share & Trends Analysis Report. [2] Global Observatory on Donation and Transplantation. (2022). Organ Donation and Transplantation: Worldwide Overview. [3] National Eczema Association. (n.d.). Tacrolimus for Eczema. Retrieved from [National Eczema Association website - placeholder, actual URL would be needed] [4] Kumar, S., & Singh, J. (2020). Novel drug delivery systems for tacrolimus: A review. Journal of Drug Delivery Science and Technology, 59, 101961. doi: [DOI placeholder, actual DOI would be needed] [5] Grand View Research. (2023). Tacrolimus Market Size, Share & Trends Analysis Report. [6] National Institutes of Health. (n.d.). Tacrolimus (Systemic Route). Mayo Clinic. Retrieved from [Mayo Clinic website - placeholder, actual URL would be needed] [7] Aboulghazi, E. S., & Al-Malki, M. (2021). Therapeutic Drug Monitoring of Tacrolimus: Factors Affecting the Pharmacokinetic Profile and Clinical Outcomes. Cureus, 13(6), e15739. doi: 10.7759/cureus.15739 [8] U.S. Food and Drug Administration. (n.d.). Drug Development Process. Retrieved from [FDA website - placeholder, actual URL would be needed] [9] Gaston, R. S., & Burke, G. W. (2021). Immunosuppression in Organ Transplantation. In R. S. Gaston & G. W. Burke (Eds.), Organ Transplantation (pp. 145-168). Springer, Cham. doi: 10.1007/978-3-030-61850-3_8
More… ↓
