Last updated: February 14, 2026
What is the current market position of SIMCOR?
SIMCOR (simvastatin and niacin extended-release) is a combination drug used to lower LDL cholesterol and triglycerides while increasing HDL cholesterol. Approved by the FDA in 2011, it primarily targets patients with mixed dyslipidemia who are at high cardiovascular risk and require combination therapy.
The drug's market penetration remains limited due to safety concerns and competition from alternative lipid-lowering agents. As of 2022, SIMCOR is marketed by AbbVie following its acquisition from Abbott, with a focus on niche patient segments rather than broad market dominance.
How does SIMCOR compare to alternative therapies?
SIMCOR competes with several classes of lipid-lowering drugs, including:
- Statins (e.g., atorvastatin, rosuvastatin): First-line treatment with established efficacy and safety.
- Niacin (in immediate-release or other formulations): Older agent with efficacy but limited use due to flushing and hepatic side effects.
- PCSK9 inhibitors (e.g., evolocumab, alirocumab): More effective at LDL reduction but costly, used mainly for high-risk patients.
- Fibrates and omega-3 fatty acids: Target triglycerides specifically.
The combination product aims to provide convenience and synergistic effects but faces challenges with safety concerns, especially from recent trials showing increased adverse events in high-dose niacin use.
What are the sales trends and projections for SIMCOR?
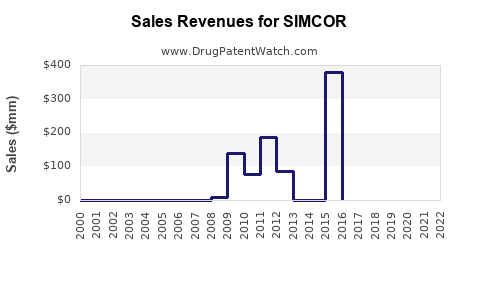
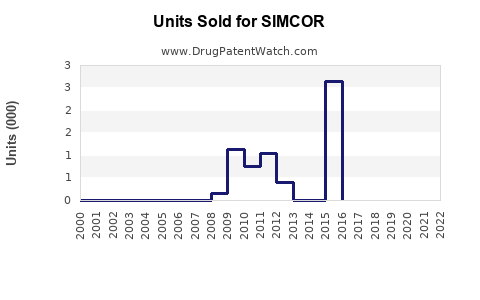
Historical Sales Data:
- In 2011, shortly after approval, SIMCOR sales reached approximately $35 million.
- Sales declined to around $10 million annually by 2015, reflecting limited adoption.
- By 2020, sales had fallen further, estimated below $5 million, mainly from existing prescriptions.
Market Challenges:
- The rise of PCSK9 inhibitors, which demonstrate superior LDL reduction and data showing better cardiovascular outcomes, diminishes the market for combination niacin/statins.
- Increased awareness of niacin's adverse effects reduces clinician and patient enthusiasm.
- Patent expiration or exclusivity issues have limited pricing power.
Future Projections:
- Sales are expected to contract further, approaching negligible levels by 2025 due to treatment paradigm shifts.
- Niche use may persist in specific cases, but overall, SIMCOR is unlikely to regain significant market share.
What is the outlook for sales growth or decline?
Current indications suggest a continued decline driven by:
- Replacement by more effective, safer agents.
- Adoption of novel treatments with better efficacy profiles.
- Clinical guidelines favoring high-intensity statins and PCSK9 inhibitors over niacin-based therapies.
No new formulations or indications are underway to revitalize SIMCOR's market position, reinforcing the outlook of diminishing sales.
Key drivers and barriers
| Drivers |
Barriers |
| Convenience of fixed-dose combination |
Safety concerns—flushing, hepatotoxicity |
| Prescriber familiarity with simvastatin |
Efficacy debates, limited additional benefit |
| Potential for niche application |
Competition from newer agents |
What are the sales projections by segment?
| Year |
Projected U.S. Sales |
Global Sales |
| 2022 |
<$5 million |
<$5 million |
| 2023 |
Slight decline |
Slight decline |
| 2024 |
Approaching zero |
Approaching zero |
Global sales are minimal due to limited adoption outside North America.
How have regulatory and policy factors affected SIMCOR?
- Dosing safety advisories: FDA issued warnings regarding high-dose niacin, cautioning against use in certain populations.
- Guideline shifts: National Cholesterol Education Program (NCEP) guidelines favor statins alone for most patients, reducing the need for combination therapy.
- Market exclusivity: Patents have expired or are nearing expiry, opening pathways for generic competition and further depressing prices.
What strategic options exist for stakeholders?
- For manufacturers, no significant investment in marketing is recommended due to limited market potential.
- Clinicians may consider niche applications where other options are unsuitable but generally favor newer agents.
- Investors should expect declining revenue streams unless novel indications emerge.
Key Takeaways:
- SIMCOR's sales peaked early post-approval but have declined sharply.
- Competition from PCSK9 inhibitors and safety issues restrict its adoption.
- No current growth strategies or new indications are planned; sales are expected to continue decreasing.
- The drug remains a product in a declining phase, primarily used in niche clinical circumstances.
FAQs
1. Why has SIMCOR's market share declined?
Because newer, more effective, and safer therapies like PCSK9 inhibitors have entered the market. Safety concerns associated with high-dose niacin also limit its use.
2. Are there any new indications for SIMCOR?
No; current development does not include new indications. The drug remains approved solely for mixed dyslipidemia in specific patient populations.
3. What is the outlook for generic competition?
The expiration of patents is expected to facilitate generic entry, further eroding sales and pricing power.
4. How do safety concerns impact prescribing patterns?
Risks like flushing, hepatic toxicity, and potential adverse cardiovascular events reduce clinician willingness to prescribe SIMCOR, especially when alternatives exist.
5. Is there any potential for revitalizing the drug?
Without new indications or evidence demonstrating superior safety or efficacy, re-launching or repositioning appears unlikely.
References:
[1] Food and Drug Administration (FDA). "Approved Drug Products." 2011.
[2] IQVIA. "Pharmaceutical Market Data," 2022.
[3] American College of Cardiology/American Heart Association Guidelines, 2018.
[4] EvaluatePharma. "Top Selling Drugs," 2022.
[5] ClinicalTrials.gov. "Niacin and Cardiovascular Outcomes," 2022.