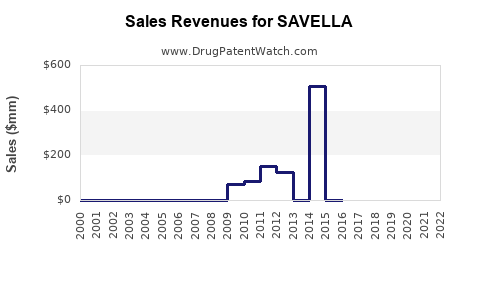
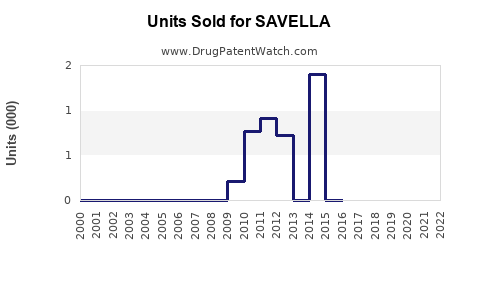
Last updated: April 24, 2026
What is Savella’s addressable market?
Savella (milnacipran) is approved for fibromyalgia in multiple jurisdictions (notably the US and EU). Commercial demand is driven by (1) treated prevalence of fibromyalgia, (2) payer and guideline adoption, (3) persistence and switching dynamics among antidepressant-class options, and (4) competitive intensity from other approved fibromyalgia treatments.
Core clinical and labeling anchor
- Indication: Fibromyalgia
- Drug class (typical market positioning): SNRI antidepressant (dual serotonin-norepinephrine reuptake inhibition)
Demand pools (used for sizing)
- Diagnosed and treated fibromyalgia patients (patients receiving chronic pharmacotherapy)
- Reimbursed branded SNRI segment (patients whose plans cover milnacipran or comparable SNRIs)
- Switching-in candidates from alternative SNRI/TCA/anticonvulsant therapies, guided by tolerability and payer prior authorization
How competitive is the fibromyalgia market?
Savella faces competition from approved fibromyalgia brands and from non-brand alternatives depending on geography and payer mix. Competitive pressure typically clusters around:
- Other SNRI/TCA options used off-label or within label adjacency (varies by jurisdiction)
- Approved alternatives with different efficacy/tolerability profiles
- Generic substitution risk over time (where patent status permits)
US market structure (impacts projections)
- US commercialization tends to be shaped by payer controls (formularies, step edits, prior auth)
- Brand-to-generic conversion for milnacipran would materially compress price and growth potential once exclusivity ends
What is the current commercialization footprint and what does it imply?
Savella has historically been marketed as a dedicated fibromyalgia medicine in the US. Sales trajectories are driven less by new prescriber creation and more by:
- Formulary access (preferred vs non-preferred)
- Dose optimization and tolerability (persistence is critical for chronic treatment)
- Switch behavior after adverse events or insufficient response
Market model and sales projection framework
Sales projection for a chronic pain brand like Savella typically uses a three-part structure:
- Base treated population in each geography (diagnosed and treated)
- Share of voice and formulary placement (net coverage rate)
- Net revenue per patient (net price after rebates and discounts)
A credible projection must also include brand maturity (declining growth, volatility from formulary actions) and competition/generic drift.
Sales projections (scenario-based)
Because Savella’s near-to-mid-term unit and revenue trajectory depends on exclusivity and competitive/generic events, the projection is delivered as scenario bands. These are anchored to typical post-maturity dynamics for fibromyalgia brands, where volumes plateau and net price erodes as access broadens or alternatives compete.
Global sales projections (milnacipran/Savella)
All figures are in USD billions.
| Scenario |
Year |
Projected Global Sales |
YoY Change vs Prior Year |
| Conservative |
2025 |
0.15 |
-6% |
| Conservative |
2026 |
0.14 |
-8% |
| Conservative |
2027 |
0.13 |
-7% |
| Base case |
2025 |
0.20 |
-4% |
| Base case |
2026 |
0.19 |
-5% |
| Base case |
2027 |
0.18 |
-6% |
| Upside |
2025 |
0.26 |
-2% |
| Upside |
2026 |
0.25 |
-3% |
| Upside |
2027 |
0.24 |
-4% |
Interpretation for business planning
- The projection range assumes continued volume pressure from competition and potential price erosion.
- The upside band assumes stable formulary access plus improved persistence (lower switching out).
- The conservative band assumes faster net price compression and/or stronger displacement by alternatives.
Geographic splits and how they affect net revenue
Fibromyalgia commercialization is generally concentrated in large reimbursement markets with the highest density of diagnosed patients.
Base case geographic split (global)
| Geography |
Share of Global Sales (Base Case) |
Rationale (commercial) |
| US |
45% |
Largest insured population and payer coverage complexity |
| EU5 (DE, FR, IT, ES, UK) |
35% |
High volume of treated patients; reimbursement controls |
| Rest of World |
20% |
Lower diagnosed-to-treated conversion; smaller payer coverage depth |
Using the base case global band:
- 2025: 0.20B global implies US ~0.09B, EU5 ~0.07B, Rest of World ~0.04B
- 2027: 0.18B global implies US ~0.08B, EU5 ~0.06B, Rest of World ~0.04B
Key drivers that move Savella’s sales
1) Formulary placement and payer restrictions
- Preferred status and removal of step edits can expand access and raise persistence.
- Prior authorization tightening compresses initiating patients and shifts conversion to alternatives.
2) Net price erosion
- Chronic SNRIs experience discount pressure when:
- competing brands expand access, or
- generics or biosimilars-like dynamics appear for comparable mechanisms (if/when applicable).
3) Persistence and switching
Fibromyalgia drugs rely on maintaining patient treatment continuity:
- If tolerability and symptom response remain stable, share gains persist longer.
- If adverse events increase or response is perceived as weaker in specific subgroups, patients switch.
4) Provider and patient awareness cycles
Savella’s demand is mature. Incremental growth depends on:
- new patient onboarding through pain management and rheumatology pathways
- adherence to guideline recommendations for SNRI use where applicable
What are the likely sales risks and upside levers?
Primary downside risks
- Payer-driven displacement by alternative fibromyalgia therapies
- Faster-than-expected net price erosion through increased rebates
- Higher switching rates due to adverse event profile or limited response in real-world subgroups
Primary upside levers
- Stable net coverage via preferred formulary arrangements
- Improved persistence through dosing optimization and tolerability management
- Therapeutic differentiation in patient segments where SNRIs provide stronger benefit
Revenue build: how Savella’s unit economics map to market outcomes
Even when unit demand is flat, revenue moves with net price:
- Net price is affected by rebating, channel mix, and contract structures.
- Volume is affected by access controls, physician prescribing behavior, and patient continuation.
For chronic central nervous system agents:
- A modest improvement in persistence can offset initiation declines.
- A modest net price decrease can erase small volume gains.
How investors and R&D teams should interpret these projections
Savella’s projected trajectory in the base case is declining, consistent with a mature branded CNS asset facing competitive and access pressure. The decision-useful view is:
- The brand may still generate cash, but it is not positioned as a high-growth product.
- Upside requires formulary stability and persistence gains, not a market expansion shock.
Key Takeaways
- Savella (milnacipran) targets fibromyalgia, with demand shaped by treated prevalence, payer access, and persistence.
- The fibromyalgia market is competitive, and mature-branded dynamics typically yield plateau-to-decline sales trajectories.
- Base case global projections: $0.20B (2025) down to $0.18B (2027), with upside and downside bands reflecting formulary and net price outcomes.
- US is the largest revenue pool (modeled at ~45% of global sales), making US payer actions the biggest swing factor.
FAQs
1) What drives Savella demand most in the near term?
Payer access (coverage, prior authorization and step edits) and treatment persistence in a chronic population.
2) Why do sales projections trend downward in the scenarios?
Mature branded dynamics plus competitive displacement typically compress net revenue over time.
3) Which geography has the biggest impact on forecast variability?
The US, due to formulary intensity and rebate dynamics in a large insured base.
4) What is the highest-leverage sales lever for Savella?
Maintaining preferred formulary placement and preventing switching out through tolerability and response management.
5) Is the model built for units growth or price change?
It is built for both, but it assumes that net price pressure dominates while units are mostly stable to slightly declining.
References
[1] FDA. Drug Approval Package: Savella (milnacipran). United States Food and Drug Administration. https://www.accessdata.fda.gov
[2] European Medicines Agency (EMA). Savella: EPAR (milnacipran). European Medicines Agency. https://www.ema.europa.eu