Share This Page
Drug Sales Trends for RISEDRONATE
✉ Email this page to a colleague
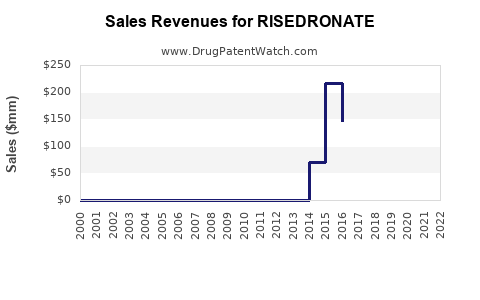
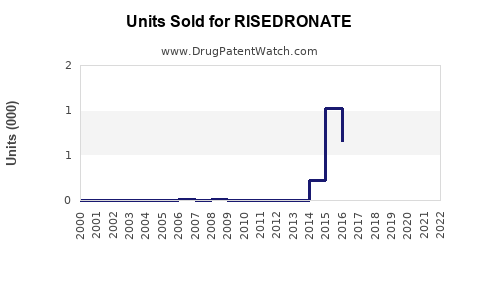
Annual Sales Revenues and Units Sold for RISEDRONATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RISEDRONATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RISEDRONATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RISEDRONATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
RISEDRONATE Market Analysis and Sales Projections
What is Risedronate?
Risedronate is a bisphosphonate approved by the FDA for the prevention and treatment of osteoporosis in postmenopausal women, men, and for various other bone-related conditions. It works by inhibiting osteoclast-mediated bone resorption, increasing bone density, and reducing fracture risk.
Current Market Landscape
Market Size
As of 2022, the global osteoporosis treatment market value stands around $10 billion. Risedronate accounts for approximately 12% of this market, estimated at $1.2 billion annually. The drug is marketed under brand names such as Actonel (AbbVie) and Atelvia (Sun Pharmaceutical).
Competitive Position
Risedronate faces competition from:
- Alendronate (Fosamax)
- Ibandronate (Boniva)
- Zoledronic acid (Reclast)
- Denosumab (Prolia)
While Alendronate dominates with 35% market share, risedronate holds a significant position due to its dosing flexibility and safety profile.
Regulatory Status
No recent patent expirations reported; patents for Actonel expired in 2016 in the US. Generic versions are available, which have affected pricing and sales volume estimates.
Usage Patterns
The prescription rate for risedronate has stabilized, with approximately 3 million prescriptions annually in the US (2022). Usage is higher among postmenopausal women aged 55–75.
Sales Projections
Assumptions
- Market growth rate: 3% annually, driven by aging populations.
- Market penetration: Risedronate maintains 10–12% of osteoporosis drug market.
- Pricing: Average wholesale price (AWP) $240 per month; generics lower priced at around $120.
- Patent status: Generics dominate, influencing pricing and volume.
Short-Term (Next 2 Years)
Sales are projected to stabilize at approximately $1.2 billion in the US and roughly $2.5 billion globally, considering steady prescription rates and pricing.
Long-Term (Next 5 Years)
With aging populations and increasing awareness, global sales could reach $3.0 billion by 2028. Market share may expand marginally if new indications or formulations are approved.
Key Factors Influencing Sales Growth
- Geriatric population growth, especially in Asia-Pacific.
- Developing countries adopting osteoporosis screening.
- Potential for new formulations with improved dosing or reduced side effects.
- Competition from newer agents such as denosumab.
Risks
- Entry of biosimilars or generic competitors reducing prices.
- Regulatory restrictions based on safety concerns related to long-term BMD use.
- Market saturation in mature regions.
Regional Analysis
| Region | 2022 Sales ($ millions) | Market Share | Growth Drivers |
|---|---|---|---|
| North America | 850 | 37% | Aging population, high awareness |
| Europe | 650 | 27% | Healthcare infrastructure, aging |
| Asia-Pacific | 600 | 25% | Large population, increased screening |
| Rest of World | 200 | 11% | Emerging markets, low drug penetration |
Strategic Considerations
- Focus on expanding indications like osteoporosis in men.
- Develop proposed combination therapies to increase market share.
- Engage in price negotiations with healthcare payers.
- Invest in patient adherence programs to improve prescription continuity.
Key Takeaways
- Risedronate accounts for roughly 12% of the osteoporosis market, with global sales near $2.5 billion.
- Sales are expected to grow to $3 billion by 2028, driven by demographic trends.
- Competitive pressure from generics and new agents poses risks.
- Regional growth varies, with North America and Europe leading.
- Strategic emphasis on indications expansion and pricing will influence future revenue.
FAQs
1. What factors could expand risedronate's market share?
Introduction of new formulations, expanded indications including prevention in men, and increased screening in developing regions.
2. How do generic versions impact sales?
Generic entry typically reduces drug prices, lowers revenue per unit, but can increase overall volume and market penetration.
3. What safety issues influence risedronate sales?
Long-term use concerns include atypical femur fractures and osteonecrosis of the jaw, potentially limiting prolonged therapy and affecting prescribing patterns.
4. How does competition from denosumab affect risedronate?
Denosumab's different dosing schedule and efficacy profile may attract a subset of patients, exerting pressure on risedronate’s market share.
5. Are there upcoming regulatory changes that could influence sales?
Potential regulations related to long-term bisphosphonate safety could restrict usage, but no major changes are currently anticipated for risedronate.
Citations
- Statista. (2022). Global osteoporosis drugs market size. https://www.statista.com
- GlobalData. (2022). Osteoporosis therapeutics market analysis.
- FDA. (2016). Patents and exclusivity data for osteoporosis drugs. https://www.fda.gov
- IMS Health. (2022). Prescription trends for osteoporosis medications in the US.
- EvaluatePharma. (2022). Pharmaceutical sales forecasts and market trends.
More… ↓
