Last updated: February 13, 2026
What is the current market size for progesterone drugs?
The global market for progesterone drugs was valued at approximately $1.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of roughly 6% through 2030, reaching around $2.0 billion.
Major markets include the United States, Europe, and Asia-Pacific. The United States accounts for approximately 45% of the current market value, driven by high infertility treatment adoption rates and hormone replacement therapy (HRT) use. Europe follows at about 30%, with expanding reproductive health markets in parts of Asia-Pacific.
How is the market segmented?
By Formulation
- Vaginal gels and creams: 40%
- Injectables: 35%
- Oral capsules: 20%
- Transdermal patches: 5%
Oral and injectable formulations currently dominate due to established prescribing practices and manufacturing capacities.
By Application
- Hormone Replacement Therapy (HRT): 55%
- Infertility treatments: 35%
- Pregnancy support (threatened miscarriage, preterm labor): 10%
HRT remains the primary driver, especially in postmenopausal women.
By Distribution Channel
- Hospitals and clinics: 50%
- Retail pharmacies: 30%
- Online pharmacies: 20%
What are the key market drivers?
- Increased use of HRT among aging populations enhances demand.
- Growing awareness of infertility treatments.
- New formulations offering improved bioavailability and convenience.
- Regulatory approvals of generic versions lowering prices, expanding access.
- Rising investments in fertility clinics and reproductive health services globally.
What are the challenges?
- Stringent regulatory requirements delaying product approvals.
- Side effects and safety concerns limit prescription rates.
- Patent expirations leading to increased generic competition.
- Variability in reimbursement policies across regions.
Who are the leading companies?
- Watson Pharma (a division of Allergan)
- Ferring Pharmaceuticals
- Pfizer
- Mitsubishi Tanabe Pharma
- Medytox
These firms hold significant market shares, primarily through proprietary formulations and distribution networks.
What are the recent sales trends?
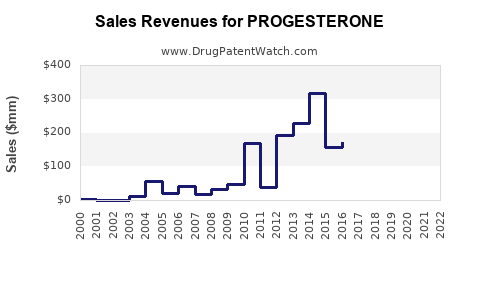
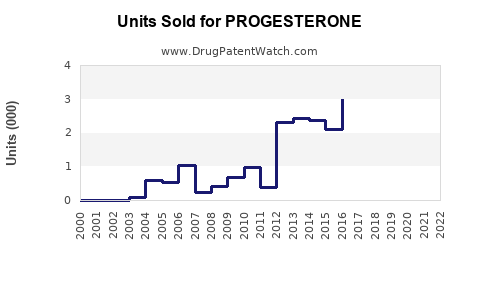
Between 2018 and 2022, global progesterone sales increased at an average of 5.5% annually. The COVID-19 pandemic initially stagnated growth in 2020 but rebounded strongly in 2021-2022 with an increased number of fertility clinics reopening and expanding services.
The U.S. market showed a 6% CAGR, outperforming Europe, with Asia-Pacific registering a 7% growth rate driven primarily by China and India.
Projected sales outlook: 2023-2030
| Year |
Global Sales (USD billion) |
CAGR |
Key factors |
| 2023 |
1.3 |
N/A |
Post-pandemic recovery, new approvals |
| 2025 |
1.7 |
6% |
Product launches, expanded reproductive health programs |
| 2030 |
2.0 |
6% |
Increasing adoption, generics penetration |
How will market dynamics change?
- Shift toward bioidentical and transdermal formulations.
- Growing prevalence of reproductive health issues in aging populations.
- Rising investments in biotech R&D, leading to novel delivery systems and indications.
- Increased regulation may slow some approvals but assure safety, fostering trust.
Key takeaways:
- The progesterone market is growing steadily, driven mainly by HRT and fertility treatments.
- Formulation innovation and generics will influence pricing and accessibility.
- The market is highly consolidated, with a few major players controlling most sales.
- Regional differences in adoption rates and regulatory policies impact sales projections.
- Future growth will depend on innovation, regulatory landscape shifts, and demographic trends.
FAQs
Q1. Which region offers the most growth potential for progesterone?
Asia-Pacific shows the highest CAGR at around 7%, driven by rising healthcare investments and increasing awareness of reproductive health.
Q2. How will patent expirations affect market competition?
Generic entrants will intensify price competition, reducing prices and expanding access but potentially reducing profit margins for branded products.
Q3. Are new formulations expected to capture significant market share?
Yes, especially bioidentical and transdermal options, which cater to patient preferences for convenience and safety.
Q4. What are main regulatory challenges?
Safety concerns, especially regarding long-term hormone use, lead to strict approval processes, which can delay market entry for new products.
Q5. What is the impact of COVID-19 on sales?
The pandemic initially hindered growth in 2020 but led to a rebound in 2021-2022, as fertility services resumed and demand increased.
References
- MarketsandMarkets. (2023). Progesterone Market by Application, Formulation, and Region - Global Forecast to 2030.
- Grand View Research. (2022). Hormone Replacement Therapy Market Size, Share & Trends.
- U.S. Food and Drug Administration. (2022). Hormone Drug Approvals and Regulations.
- Allied Market Research. (2023). Fertility Drugs Market Demand and Growth.