Last updated: February 13, 2026
What is Prevalite and Its Market Position?
Prevalite (cholestyramine) is a bile acid sequestrant used primarily to lower low-density lipoprotein (LDL) cholesterol in patients with hypercholesterolemia. It is also prescribed for treating diarrhea caused by bile acid malabsorption and to eliminate certain toxins in cases of drug overdose.
Prevalite is an established generic drug, with its patent expired in 1969. It competes mainly with other bile acid sequestrants like colesevelam and colestipol, as well as newer LDL-lowering agents such as PCSK9 inhibitors.
Market Dynamics and Key Drivers
The global hypercholesterolemia treatment market was valued at approximately $8 billion in 2022 with a projected CAGR of around 4.2% through 2030 (source [1]). The increasing prevalence of cardiovascular disease, driven by aging populations and lifestyle factors, sustains demand for lipid-lowering drugs.
The drug's low-cost profile, owing to generic availability, makes it attractive in price-sensitive markets, especially in developing countries. Prescribing patterns are influenced by guidelines favoring statins as first-line therapy, with bile acid sequestrants like Prevalite used as adjuncts or alternatives for patients intolerant to statins.
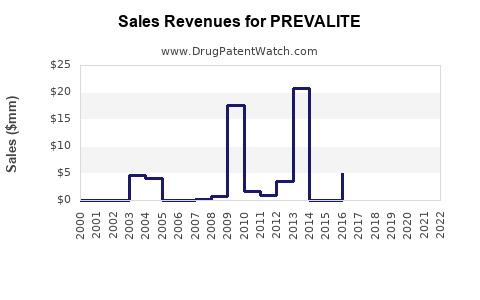
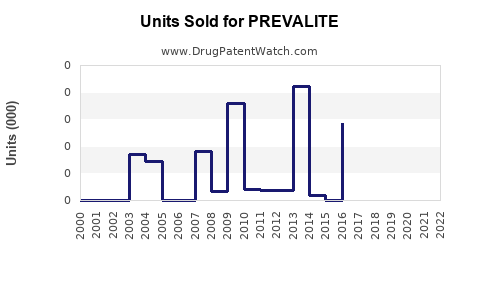
Sales Data and Historical Performance
Prevalite's sales are relatively modest compared to newer agents. In the U.S., sales peaked around 2010 at roughly $25 million annually. Since then, sales have declined due to the shift toward statins and increased use of PCSK9 inhibitors, which are more effective but costlier.
In 2021, U.S. sales were estimated at closer to $8 million, with global sales likely lower. The decline aligns with wider market trends favoring higher-efficacy treatments.
Future Sales Projection
Factors influencing future sales include:
- Market Penetration: Limited to patients with statin intolerance or hypercholesterolemia with contraindications.
- Pricing Advantage: As a generic, Prevalite remains low-cost, supporting continued use in cost-sensitive settings.
- Regulatory Changes: Any new indications or formulations could revitalize demand.
- Competitive Landscape: Dominated by newer therapies, but Prevalite can sustain niche use, especially in developing markets.
Assuming a continued decline in U.S. sales, global sales are expected to stabilize around $5–7 million annually over the next five years, primarily driven by remaining prescription volumes in cost-sensitive regions.
Market Opportunities and Risks
Opportunities:
- Expanding use in developing economies where healthcare resources favor low-cost therapies.
- Combining with other lipid-lowering agents in complex regimens.
Risks:
- Shifts toward newer, more effective therapies.
- Regulatory and safety concerns stemming from side effects like gastrointestinal discomfort and drug interactions.
- Declining physician familiarity as prescribing shifts away from older agents.
Regulatory and Market Entry Considerations
Prevalite’s generic status simplifies market access in many countries. However, its limited efficacy compared to newer agents constrains growth potential. Persuading prescribers to continue using Prevalite requires emphasizing cost benefits, especially in health systems emphasizing medication affordability.
Industry Trends and Competitive Landscape
The market for lipid-lowering drugs is increasingly dominated by:
- Statins: Remain first-line treatment with billions in sales globally.
- PCSK9 inhibitors: Generate high sales but are expensive.
- Combination therapies and novel agents: Under development, potentially reducing demand for older options like Prevalite.
Prevalite's niche remains in patients requiring non-statin options, especially where affordability is key.
Key Takeaways
- Prevalite has historically low sales, declining further as newer agents gain prominence.
- The drug's future sales hinge on its role in niche markets, chiefly in cost-sensitive settings and for specific patient populations.
- Competition from more effective therapies limits growth prospects.
- Global expansion potential exists in emerging markets, contingent on health system priorities and reimbursement policies.
- Market share retention requires emphasizing low cost and safety profiles.
FAQs
1. What are the main competitors to Prevalite?
Colesevelam and colestipol are primary competitors, along with newer LDL-lowering drugs like PCSK9 inhibitors.
2. How does Prevalite compare in efficacy to statins?
Prevalite lowers LDL cholesterol but is generally less effective and less convenient than statins, which are the first line of therapy.
3. What factors could revive Prevalite sales?
Introduction of new formulations, expanded indications, or regulatory approval for niche uses could boost sales.
4. What are the primary barriers to growth for Prevalite?
Limited efficacy, competition from more effective drugs, and declining prescribing patterns restrict growth.
5. Is there potential for market expansion in developing regions?
Yes, due to price sensitivity and healthcare budget limitations, off-label and niche use could support growth in these markets.
References
- Market Research Future, "Hypercholesterolemia Drugs Market," 2022.