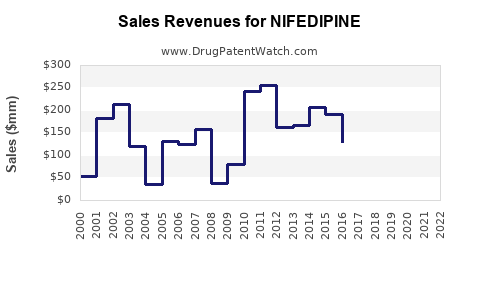
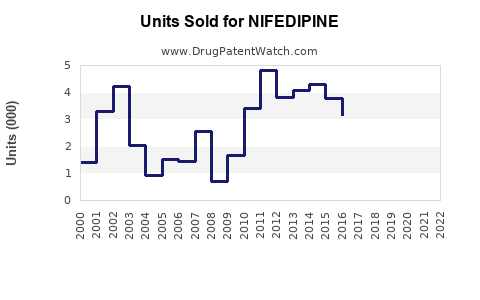
Last updated: February 12, 2026
What Is Nifedipine and How Is It Marketed?
Nifedipine is a calcium channel blocker prescribed primarily for hypertension and angina. It is available in immediate-release and extended-release formulations, marketed globally under brand names such as Adalat, Procardia, and various generics. It is classified as a prescription drug and falls under the cardiovascular agent category.
What Is the Global Market Size for Nifedipine?
The global calcium channel blocker market, which includes Nifedipine, was valued at approximately $3.4 billion in 2022. Nifedipine accounts for roughly 60% of this market, translating to an estimated $2 billion in annual sales across all formulations and regions.
How Is the Market for Nifedipine Structured?
| Segment |
Market Share |
Sales (USD billions) |
Comments |
| Immediate-release Nifedipine |
30% |
600 million |
Predominant in emerging markets |
| Extended-release Nifedipine |
70% |
1.4 billion |
Preferred in developed countries for compliance |
Market distribution indicates that the extended-release formulation dominates due to better safety profiles and patient adherence.
What Are the Regional Sales Dynamics?
| Region |
Market Share |
Estimated Sales (USD billions) |
Key Trends |
| North America |
40% |
800 million |
High penetration, extensive generic competition |
| Europe |
25% |
500 million |
Growing epidemiology, regulatory support for generics |
| Asia-Pacific |
25% |
500 million |
Rapid market growth, high prevalence of hypertension |
| Rest of World |
10% |
200 million |
Emerging markets, pricing pressures |
North America holds the largest share, driven by widespread use in hypertension management. The Asia-Pacific region demonstrates the fastest growth, attributed to increasing cardiovascular disease incidence and healthcare infrastructure expansion.
Which Factors Drive Future Sales of Nifedipine?
- Demographic Trends: Aging populations in developed regions increase demand for antihypertensives.
- Prevalence of Cardiovascular Diseases: Rising hypertension rates in emerging markets expand market size.
- Generic Market Penetration: Patent expirations in key markets lead to price competition but broaden access.
- Formulation Innovations: New extended-release formulations improve compliance, boosting sales.
- Regulatory Environment: Approvals by agencies such as FDA and EMA facilitate market expansion.
What Are Future Sales Projections Through 2030?
Based on compound annual growth rate (CAGR) estimates:
- The overall calcium channel blocker segment is projected to grow at 4% annually.
- Nifedipine sales are expected to increase from $2 billion in 2022 to approximately $2.5 billion by 2030.
This projection assumes steady growth driven by increased hypertension prevalence, continued market penetration, and ongoing generic competition. The Asia-Pacific market is anticipated to lead growth, with CAGR reaching 6% due to rising healthcare investments.
What Opportunities and Risks Exist in the Nifedipine Market?
Opportunities:
- Expansion into emerging markets with rising cardiovascular disease frequency.
- Development of sustained-release formulations with improved delivery mechanisms.
- Strategic partnerships with regional manufacturers to penetrate local markets.
Risks:
- Patent expirations may lead to price erosion.
- Competition from other antihypertensive classes such as ACE inhibitors and ARBs.
- Regulatory hurdles affecting new formulations or indications.
- Adverse event concerns, notably related to sudden blood pressure drops.
Key Takeaways
Nifedipine remains a leading antihypertensive, with global sales around USD 2 billion. The market benefits from aging populations, rising disease prevalence, and formulation innovations. The Asia-Pacific region exhibits higher growth potential, while North America dominates current sales due to established prescribing practices. Future growth hinges on market expansion in emerging regions, generic competition management, and formulation improvements. The projected compound annual growth rate suggests steady but moderate sales increases through the decade.
FAQs
1. How significant are patent expirations for Nifedipine?
Patent expirations have led to increased generic entries, resulting in significant price competition but also broader access.
2. What are key competitors to Nifedipine?
Other antihypertensive classes, such as ACE inhibitors (e.g., lisinopril) and ARBs (e.g., losartan), are alternatives with similar indications.
3. How are regulatory agencies influencing Nifedipine sales?
Regulatory approval for new formulations or indications can expand markets, while hurdles or bans on specific formulations (e.g., immediate-release in some countries) may restrict sales.
4. What demographic shifts are most impacting the market?
Aging populations worldwide increase hypertension prevalence, leading to higher Nifedipine usage.
5. What innovations could shape the future market?
Development of controlled-release delivery systems, combination therapies, and personalized medicine approaches are key innovation areas.
Sources:
- MarketsandMarkets. "Calcium Channel Blockers Market." 2022.
- GlobalData. "Pharmaceutical Market Analysis: Antihypertensive Drugs," 2022.
- IQVIA. "Global Prescription Drug Sales Data," 2022.
- World Health Organization. "Hypertension Fact Sheet," 2021.
- U.S. Food and Drug Administration. "Generic Drug Approvals," 2022.